Cardiac Computed Tomography
Emerging Cardiac Devices and Technologies
Recent studies have confirmed that non-invasive coronary imaging using Computed Tomographic Coronary Angiography (CTCA) is exceptionally accurate and at the same time, compared with its invasive counterpart, is faster, cheaper and safer.
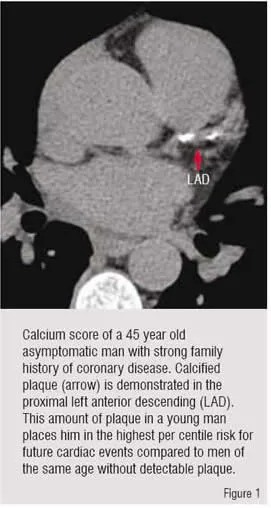
Over the last five years, the speed and resolution of mutislice Computed Tomography (CT) has advanced to the point where it can now be used for accurate cardiac evaluation1. Calcium scoring, a popular method of cardiac evaluation using Electron Beam CT technology (EBCT), has been re-evaluated using spiral CT technology, and has been validated for risk assessment of patients’ long-term cardiovascular outcomes and in varied patient ethnicities2-4. Calcium scoring, a gated non-contrast examination of the heart using cardiac CT, is a quick and safe way of evaluating for the presence of calcified atherosclerotic coronary plaque. The largest drawback of calcium score is that while a positive test confirms the process of atherosclerosis, a negative score does not completely rule out the disease. While high calcium scores can assess general risk of coronary events, they cannot accurately predict individual coronary stenosis1,5(Figure 1).
Multislice Computed Tomographic Coronary Angiography (CTCA), unlike calcium scoring, allows for the evaluation of the soft as well as the calcific plaque within the coronaries. The technique is similar, but the addition of iodinated contrast during the scan, and higher resolution imaging allows for more anatomic evaluation of the coronary structures and the surrounding cardiac anatomy5(Figure 2).
Prior to the advent of 16 slice scanners, it was difficult to evaluate the heart, much less the coronary arteries, due to the constant motion of the beating heart. With a 16-slice scan, patients would typically have to hold their breath for 25 to 30 seconds for the entire heart to be imaged. This was difficult for some patients to do, and there was acceleration of heart rates towards the end of the scan. In addition, it was difficult for the patient to remain movement free for such a long time.
Sixty-four slice scanners, which were largely available starting in 2006, allowed for acquisition of heart scans in only few seconds, and at higher heart rates than were previously possible. With the larger (approximately 4 cm) arrays in 64-slice scanners, the heart could typically be scanned in its entirety within five or six heart beats. This eliminated almost all of the problems with breath holding, and there was a much shorter time that patients needed to remain motionless. The additional benefit was that a wider range of heart rates were acceptable for scanning.
There has been a great deal of publication on 64-slice scanners with regards to accuracy of the evaluation of coronary disease, as well as its utility in evaluating the larger structures of the heart. The majority of recent evaluations suggest that the sensitivity and specificity for 64-slice scanners is exceptionally high, typically in the 90-95% range. Currently, most research and clinical trials are being performed on 64-slice technology, and the majority of scans being performed clinically are using this technology.
From a patient’s perspective, having a CTCA or calcium score is a relatively simple and painless process. Calcium scoring is slightly less involved than a CTCA, since no contrast is used, and no intravenous access is necessary. ECG leads are placed on the patient’s chest, and a 2.5 to 3mm thickness scan is performed through the level of the heart, gated to the patient’s heart beat. There is no post-imaging recovery time, and the patients are able to leave immediately. A preliminary report is often given to the patients before they are discharged. The final report is generated only after the review of the final images by the reading physician. Images are sent to a workstation for initial processing by the imaging technologist and then reviewed by the reading cardiologist or radiologist. For patients, the total time this process takes, including paperwork1, is less than 30 minutes.
For patients receiving a CTCA, the process is similar. Outpatients are typically given oral beta blockers two to three days prior to and on the day of the scan. They are asked not to eat six hours prior to scan. Once in the imaging suite, an intravenous line is placed in an antecubital vein. Telemetry is connected to ECG leads which are placed on the chest, but as lateral as possible to avoid interference with the scan. Patients who have significant renal insufficiency are typically not candidates for this test, although a mild amount of renal insufficiency is not enough to disqualify them. With 64-slice scanners, there is less of an issue related to heart rate, and patients with heart rates up to 80 bpm are still able to be imaged with excellent image quality. Patients with irregular heart rates (e.g. atrial fibrillation or a large amount of ectopy) are not good candidates for coronary imaging, although it is still feasible to evaluate the larger structures of the heart.
The CTCA scan itself consists of a series of pictures including an optional initial calcium score. A scout scan it used to localise the borders of the heart, and then a test bolus (typically 20 cc of contrast material) is used to localise the left main coronary and estimate the transit time of the contrast from the IV to the coronary arteries. The full bolus (typically 80 cc of contrast material) is injected at a rate of 3 to 5 cc / second. The patient is asked to hold their breath for the duration of the scan, which lasts 5 to 10 seconds, depending of the area of the scan. Once the scan is complete, the patient is allowed to leave. Images are sent to the reading workstation, and the reading physician evaluates the raw data, reconstructed images, and generates a final report. For patients, the process (including paperwork) takes less than one hour.

In comparison with its invasive counterpart, CTCA is much safer. The only risks are related to the contrast dye, which is potentially toxic to the kidneys, and the theoretical risk of the dose of ionising radiation. For patients with normal renal function, the contrast load is very safe. Only about 100 to 120 cc of contrast is given during a typical cardiac CT. The radiation is variable, but typically ranges from 10 to 16 mSv for a gated study6,7.
The application for multislice cardiac CT generally falls into two categories:
Evaluation of the coronaries and evaluation of the non-coronary cardiac structures (Table 1). The evaluation of coronaries is mostly done to the patients who suffer from chest pain and have a low to moderate risk of flow limiting coronary disease. It is always better to send patient for an invasive angiogram if there is a very high pre-test probability of significant coronary disease. Many patients sent for CTCA have had a prior stress test which was thought equivocal or negative, but in patients who had an invasive angiogram it is deemed overly aggressive. In this subset of patients, CTCA can define those patients in whom it is unnecessary to proceed with further testing8.
In high-risk asymptomatic patients, there is still potential use for CT to identify patients who have very premature or sub-clinical atherosclerosis9. This type of evaluation is currently not being reimbursed by insurance companies, and is often paid for by patients out-of-pocket.
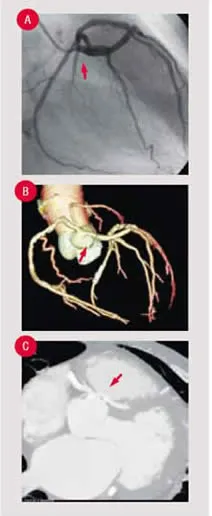
Coronary anomalies, while uncommon, are potentially lethal. Certain subsets of these patients are at high risk for sudden cardiac death, and so it is helpful to evaluate the course of the anomalous arteries (image-anomaly Figure 3) CTCA is considered the gold standard for the evaluation of the coronary tree for the presence of anomalous coronaries10. While this is its strongest indication, these patients represent a much smaller group than the patients who are suspected of having flow-limiting coronary disease.
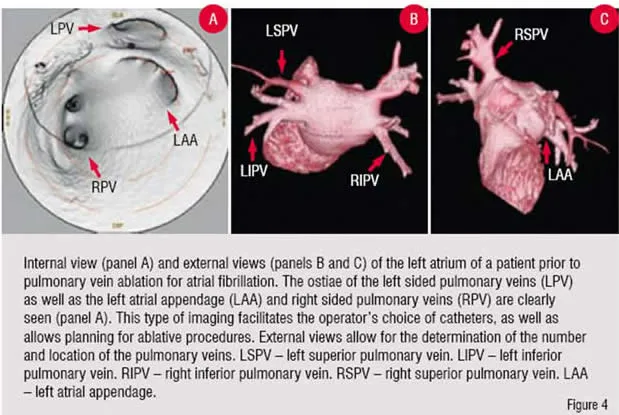
A large group of patients are now receiving cardiac scans as part of the preoperative evaluation for electrophysiology procedures such as pulmonary vein ablations for atrial fibrillation (Figure 4). In our centre, as with many others, this is becoming the standard of care prior to this type of invasive treatment11,12. A growing number of patients in whom re-operative coronary artery bypass is being contemplated are receiving CTCA to evaluate patency and location of grafts (both readily determined utilising CT) prior to repeat median sternotomy13-15. Some patients are now receiving CTCA instead of invasive angiography prior to non-coronary cardiac surgery in whom the pre-test probability of disease is relatively low16-18. The most significant change from 16 to 64-slice scanners was the reduction of acquisition time, while the resolution of the scans was essentially unchanged. Beta blockers, which are less necessary with 64-slice scanners, are still typically used as they improve the overall image quality and allow for larger radiation dose modulation.
Recently, there has been an increase in the awareness of the potential dangers of radiation, and in particular those theoretical dangers associated with ionising radiation from the increase in utilisation of computed tomography19,20. While the dosage of CTCA is higher than that of a typical invasive angiogram, the one-time dosage of a CTCA is negligible in the total impact of clinical incidence of cancer21.
There have been advances in reducing the amount of radiation by reducing the amount of peak radiation during those parts of the cardiac cycle which is less useful to image (e.g. systole). Dose modulation has been available for several years, but the most recent generation of software and hardware allows a significant dosage reduction22.
New hardwares have focussed on two areas: array size and spatial resolution. Several companies have produced scanners with larger physical arrays, which can theoretically image the heart in a single heart beat. Some of the early literature suggests that utilising a higher-slice scanner would allow for faster acquisition and reduction of additional dose23-25 and gathering more data in less time. There is almost no clinical data out on the 320-slice version of these scanners26 and at present, only a handful of clinical sites are in operation. It is likely that with the quicker acquisition times, it will be easier to scan patients whose heart rates are higher, and possible that patients with irregular heart rhythms may be imaged more successfully.
Other companies have focussed on improving detector quality and resolution, keeping the physical size of their detectors unchanged. These efforts will further improve the evaluation of plaque distribution, lumen size, and their potential ability to determine ‘at risk’ or ‘unstable’ plaque characteristics. For example, arterial collaterals, which have previously been too small to define, are potentially visible through this technology.
With these evolutionary advances in technology, there will be expansion of applications which will be seen in the coming years. One of the areas which may benefit immensely from this technology is the emergency department. Recent studies have shown that chest pain triage is feasible and potentially cost-reducing in the emergency setting27,28. It is notable that several hurdles (including rigorous patient selection) need to be addressed before this technology is utilised in a wider clinical setting29.
Calcium scoring may be supplanted by CTCA once technology to reduce overall radiation dosage is adopted. It is not too farfetched to foresee a time when an ‘at-risk’ individual will receive a screening CTCA at age 40, much in the way that certain individuals receive a screening colonoscopy.
Once the resolution of CTCA approaches that of invasive heart catheterisation (approximately 0.2 mm), there may be far fewer diagnostic procedures performed. Besides preoperative scans performed in patients with low-risk for flow limiting coronary disease, it may become feasible to scan individuals who are high-risk for left main30 and multivessel disease, sending them to coronary bypass without having an invasive catheterisation. It will also be more feasible to visualise post-stent patients to evaluate for in-stent restenosis31,32. If this becomes the norm, there may be shift in training programmes away from producing ‘invasive’ cardiologist who do not perform interventions. Resource utilisation may change, with a greater focus on outpatient diagnostic imaging with a larger percentage of patients being referred directly for intervention or surgery.
Overall, the future of cardiac computed tomography looks bright. There is a strong advocacy for the adoption of this technology. Active training programmes are being conducted throughout the world, with the first board certification in 2008. The technology is proven even in its infancy, and appears to be here to stay. Cardiac computed tomography, which has advanced quickly in only a few years, stands poised for more evolutionary leaps over the next decade, as it is incorporated into our cardiovascular treatment algorithms.
Current Indications for Cardiac Computed Tomography
• Chest pain in patients with moderate risk for flow limiting coronary disease
• Chest pain in patients with equivocal stress testing
• Coronary anomalies
• Cardiac evaluation prior to pulmonary vein ablation
• Post coronary bypass evaluation of graft patency
Possible Future Indications for Cardiac Computed Tomography
• Screening for early presence of asymptomatic coronary disease
• Pre-operative coronary evaluation for non-coronary cardiac surgery
• Pre-operative coronary evaluation for coronary bypass
• Pre-operative coronary evaluation for non-cardiac surgery
• Post-PCI evaluation of coronary stent patency`
References
1. Schussler JM, Dockery WD, Moore TR, Johnson KB, Rosenthal RL, Stoler RC. Computed tomographic coronary angiography: experience at Baylor University Medical Center/Baylor Jack and Jane Hamilton Heart and Vascular Hospital. Proc (Bayl UnivMed Cent) 2005;18:228-33.
2. Brown ER, Kronmal RA, Bluemke DA, Guerci AD, Carr JJ, Goldin J, Detrano R. Coronary calcium coverage score: determination, correlates, and predictive accuracy in the Multi-Ethnic Study of Atherosclerosis. Radiology 2008;247:669-75.
3. Ho JS, Fitzgerald SJ, Stolfus LL, Wade WA, Reinhardt DB, Barlow CE, Cannaday JJ.
Relation of a coronary artery calcium score higher than 400 to coronary stenoses detected using multidetector computed tomography and to traditional cardiovascular risk factors. Am J Cardiol 2008;101:1444-7.
4. LaMonte MJ, FitzGerald SJ, Church TS, Barlow CE, Radford NB, Levine BD, Pippin JJ, Gibbons LW, Blair SN, Nichaman MZ. Coronary artery calcium score and coronary heart disease events in a large cohort of asymptomatic men and women. Am J Epidemiol 2005;162:421-9.
5. Schussler JM, Grayburn PA. Non-invasive coronary angiography using multislice computed tomography. Heart 2007;93:290-7.
6. Dill T, Deetjen A, Ekinci O, Mollmann S, Conradi G, Kluge A, Weber C, Weber M, Nef H, Hamm CW. Radiation dose exposure in multislice computed tomography of the coronaries in comparison with conventional coronary angiography. Int J Cardiol 2008;124:307-11.
7. Coles DR, Smail MA, Negus IS, Wilde P, Oberhoff M, Karsch KR, Baumbach A. Comparison of radiation doses from multislice computed tomography coronary angiography and conventional diagnostic angiography. J Am Coll Cardiol 2006;47:1840-5.
8. Hendel RC, Patel MR, Kramer CM, Poon M, Hendel RC, Carr JC, Gerstad NA, Gillam LD, Hodgson JM, Kim RJ, Kramer CM, Lesser JR, Martin ET, Messer JV, Redberg RF, Rubin GD, Rumsfeld JS, Taylor AJ, Weigold WG, Woodard PK, Brindis RG, Hendel RC, Douglas PS, Peterson ED, Wolk MJ, Allen JM, Patel MR. ACCF/ACR/SCCT/SCMR/ASNC/NASCI/SCAI/SIR 2006 appropriateness criteria for cardiac computed tomography and cardiac magnetic resonance imaging: a report of the American College of Cardiology Foundation Quality Strategic Directions Committee Appropriateness Criteria Working Group, American College of Radiology, Society of Cardiovascular Computed Tomography, Society for Cardiovascular Magnetic Resonance, American Society of Nuclear Cardiology, North American Society for Cardiac Imaging, Society for Cardiovascular Angiography and Interventions, and Society of Interventional Radiology. J Am Coll Cardiol 2006;48:1475-97.
9. Romeo F, Leo R, Clementi F, Razzini C, Borzi M, Martuscelli E, Pizzuto F, Chiricolo G, Mehta JL. Multislice computed tomography in an asymptomatic high-risk population. Am J Cardiol 2007;99:325-8.
10. Berbarie RF, Dockery WD, Johnson KB, Rosenthal RL, Stoler RC, Schussler JM. Use of multislice computed tomographic coronary angiography for the diagnosis of anomalous coronary arteries. Am J Cardiol 2006;98:402-6.
11. Tops LF, Schalij MJ. Multislice CT: is it essential before atrial fibrillation ablation? Heart 2008;94:973-5.
12. Martinek M, Nesser HJ, Aichinger J, Boehm G, Purerfellner H. Impact of integration of multislice computed tomography imaging into three-dimensional electroanatomic mapping on clinical outcomes, safety, and efficacy using radiofrequency ablation for atrial fibrillation. Pacing Clin Electrophysiol 2007;30:1215-23.
13. Chiurlia E, Menozzi M, Ratti C, Romagnoli R, Modena MG. Follow-up of coronary artery bypass graft patency by multislice computed tomography. Am J Cardiol 2005;95:1094-7.
14. Rossi R, Chiurlia E, Ratti C, Ligabue G, Romagnoli R, Modena MG. Noninvasive assessment of coronary artery bypass graft patency by multislice computed tomography. Ital Heart J 2004;5:36-41.
15. Demaria RG, Vernhet H, Battistella P, Frapier JM, Rouviere P, Albat B. Off-Pump Coronary Artery Bypass Grafts Assessment by Multislice Computed Tomography. Heart Surg Forum 2004;7:51-54.
16. Tandon A, Allison RB, Grayburn PA, Hamman BL, Schussler JM. Preoperative visualization of a muscular ventricular septal defect by 64-slice cardiac computed tomography. Proc (Bayl Univ Med Cent) 2008;21:281.
17. Gibbs WN, Hamman BL, Roberts WC, Schussler JM. Diagnosis of congenital unicuspid aortic valve by 64-slice cardiac computed tomography. Proc (Bayl Univ Med Cent) 2008;21:139.
18. Berbarie RF, Aslam MK, Kuiper JJ, Matter GJ, Martin AW, Roberts WC, Schussler JM. Preoperative exclusion of significant coronary artery disease by 64-slice CT coronary angiography in a patient with a left atrial myxoma. Proc (Bayl Univ Med Cent) 2006;19:121.
19. Brenner DJ, Hall EJ. Computed tomography--an increasing source of radiation exposure. N Engl J Med 2007;357:2277-84.
20. Brenner DJ, Sachs RK. Estimating radiation-induced cancer risks at very low doses: rationale for using a linear no-threshold approach. Radiat Environ Biophys 2006;44:253-6.
21. Brenner DJ, Doll R, Goodhead DT, Hall EJ, Land CE, Little JB, Lubin JH, Preston DL, Preston RJ, Puskin JS, Ron E, Sachs RK, Samet JM, Setlow RB, Zaider M. Cancer risks attributable to low doses of ionizing radiation: assessing what we really know. Proc Natl Acad Sci U S A 2003;100:13761-6.
22. Abada HT, Larchez C, Daoud B, Sigal-Cinqualbre A, Paul JF. MDCT of the coronary arteries: feasibility of low-dose CT with ECG-pulsed tube current modulation to reduce radiation dose. AJR Am J Roentgenol 2006;186:S387-90.
23. Funabashi N, Mizuno N, Yoshida K, Tsunoo T, Mori S, Tanada S, Endo M, Komuro I. Superiority of synchrony of 256-slice cone beam computed tomography for acquiring pulsating objects. Comparison with conventional multislice computed tomography. Int J Cardiol 2007;118:400-5.
24. 256-slice wide-detector computed tomography. Health Devices 2007;36:362-7.
25. Mori S, Endo M, Nishizawa K, Murase K, Fujiwara H, Tanada S. Comparison of patient doses in 256-slice CT and 16-slice CT scanners. Br J Radiol 2006;79:56-61.
26. Dewey M, Zimmermann E, Laule M, Rutsch W, Hamm B. Three-vessel coronary artery disease examined with 320-slice computed tomography coronary angiography. Eur Heart J 2008;29:1669.
27. Gallagher MJ, Raff GL. Use of multislice CT for the evaluation of emergency room patients with chest pain: the so-called "triple rule-out". Catheter Cardiovasc Interv 2008;71:92-9.
28. Olivetti L, Mazza G, Volpi D, Costa F, Ferrari O, Pirelli S. Multislice CT in emergency room management of patients with chest pain and medium-low probability of acute coronary syndrome. Radiol Med (Torino) 2006;111:1054-63.
29. Schussler JM, Smith ER. Sixty-four-slice computed tomographic coronary angiography: will the "triple rule out" change chest pain evaluation in the ED? Am J Emerg Med 2007;25:367-75.
30. Schussler JM, Dockery WD, Johnson KB, Rosenthal RL, Stoler RC. Critical left main coronary artery stenosis diagnosed by computed tomographic coronary angiography. Proc (Bayl Univ Med Cent) 2005;18:407.
31. Cademartiri F, Schuijf JD, Pugliese F, Mollet NR, Jukema JW, Maffei E, Kroft LJ, Palumbo A, Ardissino D, Serruys PW, Krestin GP, Van der Wall EE, de Feyter PJ, Bax JJ. Usefulness of 64-slice multislice computed tomography coronary angiography to assess in-stent restenosis. J Am Coll Cardiol 2007;49:2204-10.
32. Martuscelli E, Razzini C, D'Eliseo A, Di Luozzo M, Mauro B, Romeo F. In-stent restenosis and multislice computed tomography: is the method ready to start? J Cardiovasc Med (Hagerstown) 2007;8:377-80.



