Heart failure management
Monitoring with implantable devices
Implantable devices, including pacemakers, defibrillators and cardiac resynchronisation systems, may play a role in helping monitor the progress of heart failure in an individual patient.
Long-term management of patients with Congestive Heart Failure (CHF) is a growing burden on healthcare systems throughout the world today. Management of patients with CHF is confounded by the observation that signs and symptoms in many of these patients are not well-correlated with actual disease status.
Hemodynamic monitoring
Current methods for routine hemodynamic evaluation (e.g. Swan-Ganz catheterisation and echocardiography) are costly and not well-suited for repeated serial measurement in the ambulatory setting. Recently, considerable investigation has focussed on the development of alternative methods of assessing patient’s disease status. Implantable hemodynamic sensors may enable frequent monitoring of hemodynamic changes in CHF patients and be used as a surrogate for serial invasive catheterisations in tailoring and titrating medical therapy. Furthermore, continuous monitoring of hemodynamic measurements might provide unique insight regarding pathophysiological mechanisms and chronic responses to treatment regimens.
Pressure monitoring system
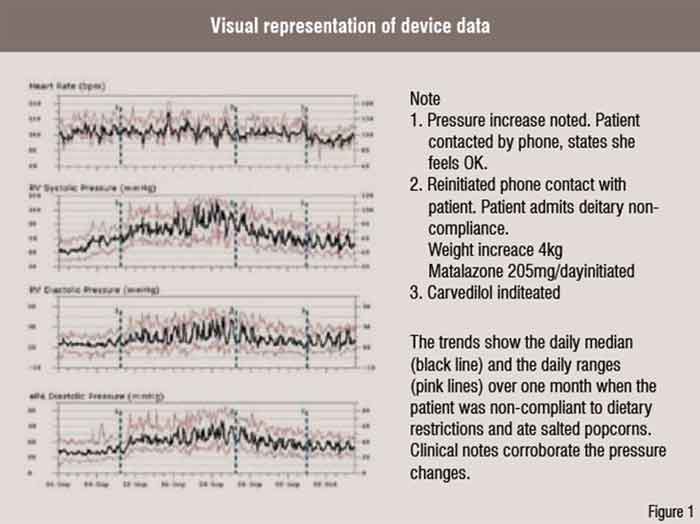
A totally Implantable Hemodynamic Monitor (IHM) has been developed for outpatient HF management. The IHM system consists of a pacemaker-like device that processes and stores information and a transvenous lead incorporating a high fidelity pressure sensor near its tip. The implantation procedure is similar to that of a single chamber pacemaker system with the lead positioned in the right ventricular outflow tract or high right ventricular septum in an area of high blood flow. The IHM measures and stores heart rate, patient activity, right ventricular systolic and diastolic pressure, estimated pulmonary arterial diastolic pressure (ePAD), positive and negative dP / dt, right ventricular pre-ejection and systolic time intervals, and body temperature. A strong correlation (r=0.84) was demonstrated between actual pulmonary artery pressures and ePAD under a variety of physiologic conditions. The IHM system continuously measures and stores hemodynamic information that can be reviewed remotely via the Internet. The website automatically processes and concatenates new data received from the device with data from previous transmissions and provides visual representation of the data in the form of trends over time (Figure 1).
Clinical trials
Several studies demonstrated safety and accuracy of the implantable hemodynamic monitoring system. The COMPASS-HF (Chronicle Offers Management to Patients with Advanced Signs and Symptoms of Heart Failure) study randomised 274 NYHA Class III-IV patients, all of whom received an IHM, to the Chronicle-guided management group (n=134) or control group (n=140) over a 6-month follow-up period. The study demonstrated the IHM was safe and able to reduce the rate of heart failure-related events. However, this 21 per cent reduction in events did not reach statistical significance (p=NS) . Retrospective analyses from COMPASS-HF provided new insights to the pathophysiology of the transition from stable, compensated HF to the decompensated state in HF patient with reduced left ventricular ejection fraction (LVEF) and among HF patients with preserved LVEF (i.e. diastolic heart failure). These data suggest that chronic management of HF by the IHM may also be comparable in these two groups of HF patients. Another currently on-going trial (Reducing Events in Patients with Chronic Heart Failure; REDUCEhf ) will prospectively test the hypothesis that the ambulatory hemodynamic monitoring can indeed reduce the rate of heart failure-related hospitalisation.
 Intrathoracic Impedance Monitoring
Intrathoracic Impedance Monitoring
The correlation between changes in biological impedance and physiologic parameters, such as respiration rate and cardiac hemodynamics has been studied for decades. For example, the estimation of left ventricular volume using the impedance catheter technique is the gold standard of physiological research quantifying left ventricular pump function since the 1980s. Likewise, many implantable devices employ impedance as a method of monitoring therapy system performance. Also, some devices use changes in impedance associated with the respiratory cycle to help automatically adjust the pacing rate. More recently, the association between acute decreases in intrathoracic impedance and acute decompensation of CHF was investigated.
Early clinical trials
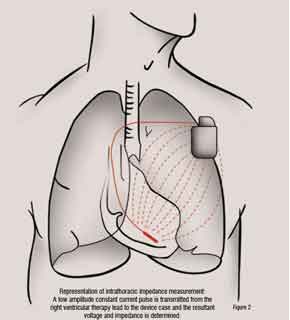
The MidHeft trial provided the first clinical evidence that daily monitoring of intrathoracic impedance measured between the right ventricular defibrillation coil and the devise case could provide a clinically useful tool for
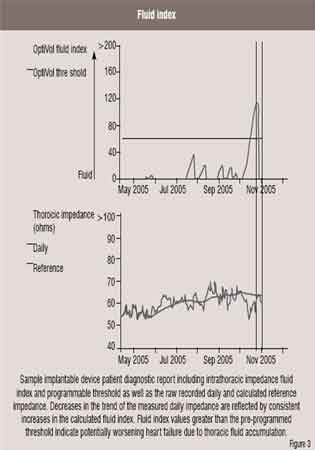
monitoring the onset of acute heart failure decompensation (Figure 2). Device recorded daily impedance data from this non-randomised double blinded prospective trial (n=33) was used to develop and validate an algorithm to detect acute pulmonary fluid accumulation based on day-to-day changes in the actual recorded daily intrathoracic impedance. The algorithm calculates dynamic reference impedance based upon trends in the measured daily intrathoracic impedance. Differences between the measured daily impedance and the calculated reference impedance are used, in turn, to increment or reset a ‘fluid index’ (Figure 3). According to the reported results of the study, the fluid index for patients who were hospitalised for acutely decompensated heart failure crossed a predetermined fluid index threshold (60 ohm days) prior to hospitalisation in over 77 per cent of the events. The changes in the calculated fluid index occurred on average 15 days prior to symptom onset. The trial also observed that the rate of fluid index threshold crossings not associated with imminent heart failure hospitalisation was about 1.5 events per patient per year. Initial results from the FAST trial, a follow-on prospective clinical trial in ICD and CRT patients, further validated the potential clinical utility of chronic intrathoracic impedance monitoring to manage heart failure.
Recent clinical findings
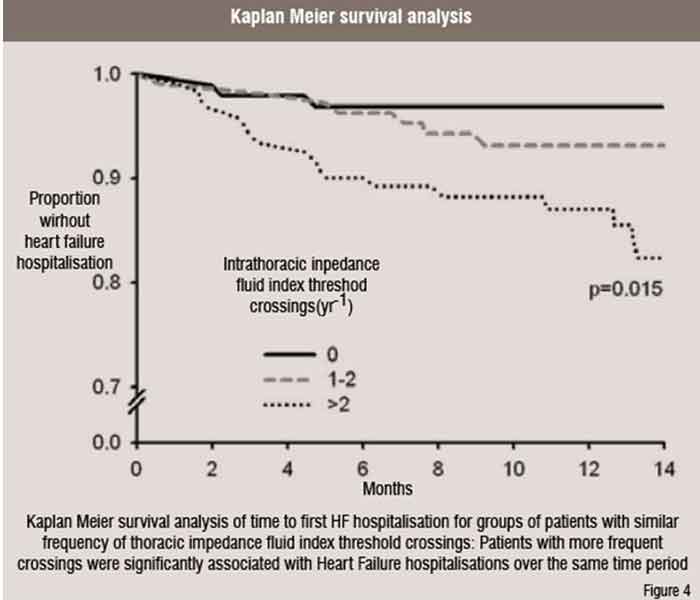
Since the MidHeft and FAST trials, a number of other published investigations further validated the clinical utility of chronic heart failure monitoring using device recorded intrathoracic impedance. Vollmann and colleagues performed a multi-centre non-randomised investigation of 372 patients with CRT-D devices that included intrathoracic impedance monitoring. Many patients in this trial were automatically alerted to fluid index threshold crossings by an audible alert tone transmitted by the device. The authors reported an adjusted sensitivity and positive predictive value of the alerts to various clinically relevant events associated with heart failure of 60 per cent respectively. Ypenburg and colleagues followed 115 CRT-D patients with intrathoracic impedance monitoring for an average of nine months. They reported that the sensitivity and specificity of the fluid index depended strongly on the programmed detection threshold. They concluded that optimal algorithm performance may require tailoring the threshold value to fit the needs of the particular patient. Small and colleagues also performed an analogous investigation in 326 US CRT-D patients with intrathoracic impedance monitoring but no audible alert, which are not yet approved for use in the US. That analysis included univariate and multivariate linear regression of changes in the fluid index as well as other device diagnostic parameters. The results indicated that each intrathoracic impedance fluid index threshold crossing was associated with a 51 per cent increased probability of a heart failure hospitalisation within the same year as the crossing (Figure 4). In addition, changes in intrathoracic impedance also showed good correlations with both weight and BNP levels. All of these trials confirmed the fundamental hypothesis that intrathoracic impedance monitoring can provide a useful clinical tool to help manage patients with congestive heart failure. However, two current randomised multi-centre clinical trials including PRECEDE HF and DOT are independently testing the hypothesis that in-office and remote monitoring of device diagnostic data, including intrathoracic impedance monitoring, will improve a combined primary endpoint of death and heart failure hospitalisation.
 Integrated diagnostics
Integrated diagnostics
Estimated filling pressure with intrathoracic impedance monitoring
Chronic monitoring of both estimated left ventricular filling pressure and Intrathoracic impedance represents the potential for further improvements and refinements to device-based remote clinical monitoring. The relationship between changes in these two parameters was investigated in a few small-scale clinical studies. These pilot data indicate good correlations between right ventricular pressure and intrathoracic impedance-derived parameters of worsening heart failure. Therefore, chronic monitoring of both estimated filling pressures as well as intrathoracic impedance may substantially improve both the sensitivity and specificity of each respective parameter to predict acutely worsening heart failure as well as other clinically relevant events prior to manifestation of clinical symptoms. Clinical trials currently underway, such as the REDUCEhf trial, should help to elucidate these complex relationships in the heart failure population. Eventually, sophisticated computational algorithms that simultaneously consider multiple parametric changes determined separately from multiple independent sensors may be developed from such clinical data.
AF / CHF
The complex relationship between CHF and both atrial and ventricular arrhythmias is well described. However, hemodynamic and electrocardiographic data recorded by implantable devices have helped to further elucidate this complex interrelationship. Recent investigations have reported intriguing correlations between changes in intrathoracic impedance and the onset of both atrial and ventricular tachyarrhythmias. These results demonstrate that reductions in intrathoracic impedance often precede individual occurrences or ‘storms’ of arrhythmias. Likewise, the onset of a persistent atrial tachyarrhythmia often portends acute decompensation for congested heart failure. The nature of this interrelationship may vary from patient to patient or perhaps even within an individual patient. Thus, implantable device diagnostics that reveal intricate temporal relationship between arrhythmia onset and acute decompensation may play a key role in tailoring therapies for both co-morbidities.
Heart rate variability
The longstanding ability of implantable devices to monitor both paced and intrinsic ventricular cycle lengths has fostered interest in leveraging that capability to monitor changes in heart rate variability and day and night heart rates. Such diagnostic data may provide relative insight into the condition of the autonomic nervous system. Several implantable devices now provide long-term diagnostic trends for such parameters. Recently, Adamson and colleagues demonstrated that reductions in device measured indices of heart rate variability often precede episodes of acute heart failure decompensation. Similarly, results of the OFISSER trial showed that increases in night heart rate were associated with acute heart failure decompensation in patients with CRT-D therapy. These diagnostic parameters are somewhat limited by other factors. For example, if the patient is experiencing a persistent atrial arrhythmia, or if the device is controlling the atrial rate by frequent pacing, then heart rate parameters such as heart rate variability are not calculated and the diagnostic information is hence unavailable. This scenario can represent the majority of time for some patients with implantable devices, especially those who are pacemaker dependent.
Arrhythmia monitoring
The ability of implantable devices to detect and discriminate various atrial and ventricular tachyarrhythmias accurately has been exhaustively documented. These capabilities are required in order to apply automatic therapies safely and appropriately for both ventricular and, in some devices, atrial tachyarrhythmias. Many devices augment these automated detection capabilities by storing ventricular and atrial bipolar electrograms recorded prior to and during tachyarrhythmias. However, besides augmenting device therapy, the ability to record and report the precise date, time duration, cycle length and example electrograms of such arrhythmias can also have important diagnostic applications. For example, it has been shown that symptoms are a relatively poor indicator of atrial tachyarrhythmia occurrence. That is, most atrial arrhythmia episodes are asymptomatic. Likewise many reported arrhythmia symptoms are not associated with an actual arrhythmia. Therefore, device monitoring capabilities represent a reliable and diligent method of monitoring arrhythmias that may bay used to help manage pharmacologic and non-pharmacological rate and rhythm control strategies. The recent TRENDS clinical trial examined the potential of these implantable device atrial arrhythmia diagnostics to help monitor the risk of thromboembolic events. This study was underpowered to achieve the primary endpoint. However, a posthoc analysis of this multi-centre non-randomised trial (n=2486) showed that the thromboembolic event risk may be a quantitative function of atrial tachyarrhythmia burden. That is, atrial tachyarrhythmia AT / AF burden greater than 5.5 hours on any of 30 days prior to the event approximately doubled the thromboembolic risk. Hence, device-detected AT / AF burden may help identify subgroups at higher risk for thromboembolic events. The ASSERT study is an even larger study with much longer follow-up and should answer many further questions regarding the predictive value of AF burden for strokes in high risk patients.
Clinical adoption and remote monitoring
The rapid advances in device monitoring capabilities have, to some extent, outstripped the ability of many clinics to leverage the plethora of available diagnostic information. However, many implantable devices now also contain telemetric capability that allows the device to transmit all sensor derived parameter trends from the patient’s home directly to the clinic. This is typically achieved through a bedside monitoring / telemetry device that can be programmed by the clinic to transmit automatically based on preset schedule. Alternatively, a transmission of device stored monitoring and diagnostic data can also be triggered based on detected clinical events. For example, some devices can be programmed to alert the clinic directly if the patient experiences the onset of atrial tachyarrhythmias or if the ventricular rate during a sustained atrial tachyarrhythmia exceeds a pre-programmed threshold. Such remote monitoring ‘care alerts’ may be quite useful to monitor rate and rhythm control strategies. These remote monitoring capabilities also foster the potential to transmit non-device recorded information automatically, such as weight, blood pressure and associated symptoms back to the managing clinic. Thus, the stage is set for a new paradigm of heart disease management based on continuous remote monitoring rather than intermittent clinic visits.
The future
The number and variety of physiologic sensors and the useful clinical parameters derived from those sensors is likely to continue to increase rapidly. For example, in addition to the capabilities described above, future devices may include additional sensors to track respiration parameters (including rate, minute ventilation and perhaps apnea and dyspnea detection), tissue perfusion (via optical sensors), cardiac output and stroke volume (via impedance acute ischemia or myocardial infarction via S-T segment monitoring), electrical alternans and heart rate turbulence. Indeed some recently released devices already contain some of these fascinating capabilities.
The development of practical chemical sensors to monitor parameters, although less mature, is also feasible. However, some external glucose pumps for the chronic management of diabetes also contain chronic intermediate-term (i.e. several days) subcutaneous glucose monitoring capability. The ability of such chemical sensors to augment other device monitoring capabilities for heart failure or other risks will require investigation.
References
Bourge RC, Abraham WT, Adamson PB, Aaron MF, Aranda Jr JM, Magalski A, Zile MR, Smith AL, Smart FW, O’Shaughnessy MA, Jessup ML, Sparks B, Naftel DL, Stevenson LW. Randomised Controlled Trial of an Implantable Continuous Hemodynamic Monitor in Patients With Advanced Heart Failure: The COMPASS-HF Study. Journal of the American College of Cardiology. 2008;51:1073-1079.
AUTHOR BIO
Michael Gold is the Michael E Assey Professor of medicine, as well as the Director of the Division of Cardiology and Associate Dean of Interdisciplinary Clinical Programs at the Medical University of South Carolina in Charleston. He has authored over 125 articles in peer-reviewed journals, in addition to over 30 book chapters. He sits on the Editorial Board of several peer-reviewed journals, including American Journal of Cardiology, PACE, Heart Rhythm, and Journal of Cardiac Electrophysiology.






