Harnessing Precision Medicine for Better Lung Cancer Screening in Asian Populations

Introduction:
Lung cancer remains the leading cause of cancer mortality globally, with a growing burden in Asian populations, especially among non-smokers and women [1]. Traditional “one-size-fits-all” screening models are insufficient in addressing the unique epidemiological, genetic, and environmental characteristics of Asian populations [2]. The prevalence of subsolid (ground-glass or part-solid) nodules differs notably between Eastern and Western populations, contributing to distinct screening challenges [3]. In Asia, the potential for overdiagnosis associated with lung cancer screening is considerable due to these imaging characteristics [4]. Furthermore, awareness and understanding of overdiagnosis vary widely among governments, healthcare institutions, medical professionals, public health researchers, and the general public [5]. These differences present challenges in implementing balanced screening policies that optimize benefits while minimizing harms. With the advent of precision medicine, lung cancer screening is undergoing a transformation toward a more personalised, predictive, and participatory approach. This article outlines a five-pillar strategy for improving lung cancer screening among Asian populations (Figure 1):
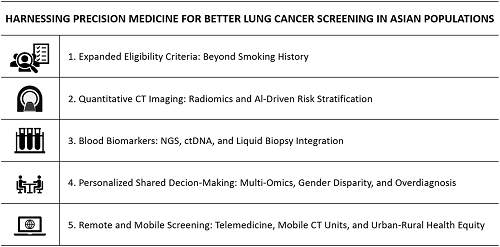
Figure 1. Harnessing precision medicine for better lung cancer screening in Asian populations: five strategic approaches.
1. Expanded Eligibility Criteria: Beyond Smoking History
While most Western guidelines restrict screening to heavy smokers, such criteria miss a substantial portion of Asian patients who develop lung cancer despite never smoking. For example, in countries like Taiwan, Japan, and South Korea, a large proportion of female patients are never-smokers [6]. Precision risk models that incorporate age, family history, environmental exposures (such as indoor air pollution or cooking fumes), and genetic susceptibility are urgently needed. These models allow for early identification of at-risk individuals regardless of smoking status.
2. Quantitative CT Imaging: Radiomics and AI-Driven Risk Stratification
Low-dose CT (LDCT) remains the gold standard for lung cancer screening, but false positives and overdiagnosis are major concerns. Emerging techniques such as radiomics and artificial intelligence (AI) allow extraction of high-dimensional features from CT images—shape, margin, density, texture, vascularity—that can be analysed to predict malignancy more accurately [7-9]. Deep learning algorithms trained on large Asian datasets can better distinguish indolent nodules from aggressive ones, improving clinical decision-making. In Asian populations, lung cancer screening must consider unique genetic and environmental factors, making precision medicine especially important. Integrating augmented artificial intelligence (AI) into low-dose computed tomography (LDCT) screening not only improves diagnostic accuracy but also reduces the workload of radiologists and enhances overall efficiency [10]. However, AI tools are a double-edged sword—when used appropriately, they offer significant screening benefits, but improper use may lead to unintended harm. Therefore, real-time guidance from experienced thoracic radiologists is crucial. These experts can mentor non-thoracic radiologists, junior doctors, and residents through AI-assisted imaging and case-based teaching, improving clinical decision-making and diagnostic confidence [11]. Yet, over-reliance on AI or misinterpretation of noisy AI-generated data can lead to diagnostic errors and flawed clinical decisions. This underscores the need for robust education, structured training, and region-specific validation protocols. Tailoring AI models to reflect the unique epidemiology and imaging characteristics of Asian populations can further optimize screening performance. By combining AI technology, precision medicine, and targeted education strategies, we can promote more equitable, efficient, and high-quality lung cancer care across Asia.
3. Blood Biomarkers: NGS, ctDNA, and Liquid Biopsy Integration
Invasive biopsies pose risks and are often infeasible for small nodules. Blood-based biomarkers, such as circulating tumor DNA (ctDNA) and circulating cDNA, offer non-invasive alternatives. Techniques like next-generation sequencing (NGS) can detect mutations (e.g., EGFR, ALK), gene expression changes, and methylation patterns. When combined with imaging findings, these markers enhance diagnostic precision and allow longitudinal monitoring. Incorporating liquid biopsy into the screening algorithm is especially valuable for resource-limited or high-risk settings [2].
4. Personalised Shared Decision-Making: Multi-Omics, Gender Disparity, and Overdiagnosis
Precision screening is not only about advanced tools, but also about aligning with patients’ values and preferences. Using multi-omics prediction models that integrate genomics, proteomics, and transcriptomics can better stratify risk. Moreover, sex-specific considerations are crucial—women may have slower-growing nodules but respond differently to interventions. Effective shared decision-making tools should incorporate overdiagnosis risk, comorbidities, psychological readiness, and gender-related prognostic models [12]. Such tools empower patients to make informed, value-congruent decisions. In this context, active surveillance may be appropriate for indolent nodules, while others may benefit from an optimised surgical timing approach tailored to individual progression risk. These strategies empower patients and clinicians to collaboratively choose management plans aligned with both clinical evidence and personal values.
5. Remote and Mobile Screening: Telemedicine, Mobile CT Units, and Urban-Rural Health Equity
Accessibility is a major challenge in Asia’s rural or underserved areas. Mobile low-dose CT trucks and telemedicine-based shared decision-making platforms can bridge geographic gaps. Patients can receive virtual risk counseling, discuss results online, and even track follow-up visits. These innovations promote urban-rural health equity and reduce disparities in screening access. Moreover, in rural settings, telehealth integration into hospital systems allows busy individuals or elderly patients to engage in screening process without transportation burdens [13]. Accessibility is a major challenge in Asia’s rural or underserved areas. Mobile low-dose CT trucks and telemedicine-based shared decision-making platforms can bridge geographic gaps [14]. Patients can receive virtual risk counseling, discuss results online, and even track follow-up visits. These innovations promote urban-rural health equity and reduce disparities in screening access. Moreover, in rural settings, telehealth integration into hospital systems allows busy individuals or elderly patients to engage in screening process without transportation burdens. Digital shared decision-making tools delivered through telehealth platforms further support patient engagement by facilitating informed, real-time discussions between patients and clinicians. This approach empowers patients to make individualised decisions that align with their unique imaging and blood biomarker profiles, clinical risks, and personal preferences.
Remote platforms can support large-scale data collection, facilitating research on lung cancer risk factors unique to Asian populations and advancing precision medicine for lung cancer in Asia. Digital shared decision-making tools delivered through telehealth platforms further support patient engagement by facilitating informed, real-time discussions between patients and clinicians. This approach empowers patients to make individualised decisions that align with their unique imaging and blood biomarker profiles, clinical risks, and personal preferences (Figure 2).

Figure 2. Tele-consultation for lung cancer screening and personalised clinical decision sharing.
Conclusion:
A Precision-Driven Future for Lung Cancer Screening in Asia
A gender-specific, precision medicine-based strategy tailored for lung cancer screening in Asian populations to enhance early detection and population relevance. For male individuals with a history of smoking, health literacy education can enhance their understanding of lung cancer risks and the benefits of screening, thereby effectively increasing the screening participation uptake rate. In contrast, for high-risk female individuals who do not smoke but have a family history of lung cancer, appropriate screening strategies can be planned through education program and shared decision-making [6]. This approach helps reduce the incidence of indolent lung cancers and overdiagnosis within the non-smoking female population. This personalised clinical information workflow—centered on sex and smoking history—enables precise risk stratification and guides each population toward the most appropriate screening pathway, facilitating effective early detection of lung cancer.
Dr. Fu-Zong Wu's team at Kaohsiung Veterans General Hospital further emphasizes the importance of precision stratification implementation into clinical practice. By integrating augmented artificial intelligence (AI) tools, they have developed a streamlined and clinically executable one-stop stratification strategy. This approach assists physicians in determining optimal management decisions—whether active surveillance or surgical intervention—for patients with pulmonary nodules, thereby enhancing the quality of clinical decision-making and overall patient care efficiency (Figure 3).
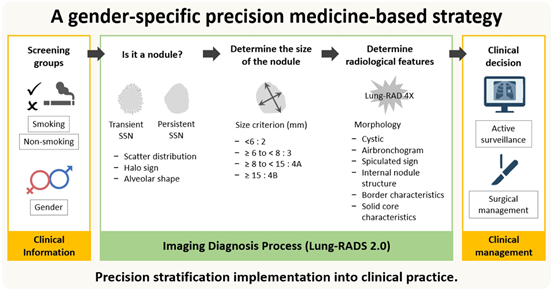
Figure 3. Developing a precision-based and personalised workflow for lung cancer screening: from criteria and Imaging to tailored clinical decision-making.
By integrating personalised risk models, AI-enhanced imaging, liquid biopsy, gender-sensitive shared decision-making, and remote/mobile technology, we can build a future-ready lung cancer screening system tailored for Asian populations (Figure 4). This comprehensive approach not only enhances early detection and minimizes the risk of overdiagnosis, but also optimizes the delicate balance between potential harms—such as unnecessary interventions, psychological distress, and radiation exposure—and the benefits of timely diagnosis and improved survival. By integrating personalised strategies at each stage of the screening process, this model supports more precise, patient-centered care. Furthermore, it advances health equity by extending access to underserved populations through telemedicine and mobile technologies, and promotes healthcare system efficiency by reducing unnecessary procedures and focusing resources on individuals with the highest risk and greatest potential to benefit.
Figure 4. Dr. Fu-Zong Wu, Associate Professor and lung cancer screening coordinator at Kaohsiung Veterans General Hospital, educates the public on CT-detected lung nodules and guides shared decision-making.
References
- Wu FZ, Huang YL, Wu CC, Tang EK, Chen CS, Mar GY, et al. Assessment of Selection Criteria for Low-Dose Lung Screening CT Among Asian Ethnic Groups in Taiwan: From Mass Screening to Specific Risk-Based Screening for Non-Smoker Lung Cancer. Clin Lung Cancer. 2016;17(5):e45-e56.
- Chen HH, Wu YJ, Wu FZ. Precision Medicine in Lung Cancer Screening: A Paradigm Shift in Early Detection-Precision Screening for Lung Cancer. Diagnostics (Basel). 2025;15(12).
- Chang YC, Hung YC, Wu YJ, Tang EK, Wu FZ. Understanding East-West differences in subsolid nodules: prevalence and overdiagnosis implications in lung cancer screening. Ann Med. 2025;57(1):2478321.
- Hsin-Hung C, En-Kuei T, Yun-Ju W, Fu-Zong W. Impact of annual trend volume of low-dose computed tomography for lung cancer screening on overdiagnosis, overmanagement, and gender disparities. Cancer Imaging. 2024;24(1):73.
- Liu YC, Liang CH, Wu YJ, Chen CS, Tang EK, Wu FZ. Managing Persistent Subsolid Nodules in Lung Cancer: Education, Decision Making, and Impact of Interval Growth Patterns. Diagnostics (Basel). 2023;13(16).
- Wu FZ, Chang YC. Toward More Effective Lung Cancer Risk Stratification to Empower Screening Programs for the Asian Nonsmoking Population. J Am Coll Radiol. 2023;20(2):156-61.
- Wu YJ, Wu FZ, Yang SC, Tang EK, Liang CH. Radiomics in Early Lung Cancer Diagnosis: From Diagnosis to Clinical Decision Support and Education. Diagnostics (Basel). 2022;12(5).
- Tang EK, Wu YJ, Chen CS, Wu FZ. Prediction of the stage shift growth of early-stage lung adenocarcinomas by volume-doubling time. Quant Imaging Med Surg. 2024;14(6):3983-96.
- Wu FZ, Wu YJ, Chen CS, Tang EK. Prediction of Interval Growth of Lung Adenocarcinomas Manifesting as Persistent Subsolid Nodules ≤3 cm Based on Radiomic Features. Acad Radiol. 2023;30(12):2856-69.
- Wu YJ, Wu FZ. AI-Enhanced CAD in Low-Dose CT: Balancing Accuracy, Efficiency, and Overdiagnosis in Lung Cancer Screening. Thorac Cancer. 2025;16(1):e15499.
- Hung YC, Lin Y, Wu FZ. Editorial: Advancements and Challenges in Lung Cancer Screening, Diagnosis, and Management. Diagnostics (Basel). 2025;15(7).
- Wu FZ, Kuo PL, Wu CC, Wu MT. The impact of patients' preferences on the decision of low-dose computed tomography lung cancer screening. Transl Lung Cancer Res. 2018;7(Suppl 3):S236-s38.
- Berzingi S, Piechowski K, Hendricks E, Colantonio M, Anandarm A, Perkowski G, et al. Integrating cardiovascular risk assessment into mobile low-dose CT lung screenings in rural Appalachia: A comprehensive analysis of the relationship between lung cancer risk, coronary artery calcium burden, and cardiovascular risk reduction strategies. Am J Prev Cardiol. 2024;19:100719.
- Tao W, Yu X, Shao J, Li R, Li W. Telemedicine-Enhanced Lung Cancer Screening Using Mobile Computed Tomography Unit with Remote Artificial Intelligence Assistance in Underserved Communities: Initial Results of a Population Cohort Study in Western China. Telemed J E Health. 2024;30(6):e1695-e704.









-(1).jpg)