
Ischemic Mitral Regurgitation (IMR) is not a valve disease in itself. It is a Functional Mitral Regurgitation (FMR) or secondary to Myocardial Infarction (MI) due to a tethering force or annular dilatation. Its incidence is approximately fifth of acute MI and half of congestive heart failure (CHF).(1) One of the mechanical complications of MI is IMR and associated with a poor prognosis. IMR is classified into acute development within a week due to rupture of the papillary muscles, and chronic develops more than a week and is associated with segmental wall motion abnormalities (SWMA). IMR mostly develops after inferior MI (38%) more than anteroseptal MI (10%).(1,2)The optimal surgical strategy for management of IMR is still debated. The objective of this study was to evaluate the early mortality and morbidity of Mitral Valve repair (MVr) in patients with IMR undergoing Coronary Artery Bypass Grafting (CABG). CABG alone has proven to be persistence MR, and poor survival. MVr or MVR can eliminate or decrease MR, and have insignificant differences in survival. However, MVr is associated with lower hospital mortality.(3,4) Management of IMR has been an issue of continued debate for several decades. The therapeutic approaches are: MVR with CABG, MVr with CABG, or CABG alone. CABG alone has proven not adequate for severe MR, with persistence MR (77%). IMR reduces survival even after CABG.(5, 6)
We performed a retrospective observational cohort study on prospectively collected data on 136 consecutive CABG patients with IMR undergoing MVr between 2016-2020. Echocardiogram findings, operative procedures, and outcomes were analyzed. MVr was performed by a downsized complete rigid annuloplasty ring as the procedure of choice. A complete annuloplasty ring decreases mitral leaflet strain, improves leaflet coaptation geometry, and enhances repair durability. The Cosgrove flexible band is designed to preserve mitral annular dynamics. It has been suggested that the saddle-shape and the sphincter mechanism of the mitral valve can be preserved for up to 5-years after implantation.
Downsizing ring was determined after careful measurement of the height of anterior mitral leaflet (AML). Rings were inserted using deep interrupted horizontal sutures using Ethibond 2-0 or Ti-Cron 2-0. MVr is performed using rigid complete ring (Carpentier-Edwards Physio).In (14) cases, posterior mitral leaflets are partially tethered to annulus and require release of secondary chordae along the length of annulus. Only in (2) cases, Papillary Muscle (PM) approximation or realignment was performed by 2-3 mattress 3/0 polypropylene sutures with PTEE pledget. Approximation was done between anterior and posterior PM in their muscle portion to come into contact with each other.
Results
Table (1): Baseline characteristics and preoperative data.
| Variable | Descriptive (n=136) |
| Age (years) | 62.3±9.8 ( 16-91) |
| Sex | Male 95(69.9%) Female 41(3.1%) |
| Total hospital stay (day) | Mean±SD (range) 19.35±22 Median 12.8 |
| ICU stay (day) | Mean ± SD ( range) 13.6±18.9 Median 8.5 |
| Operative Status | Elective 73 (53.7%) Urgent 63 (46.3%) |
| NYHA Class | Class I 4(2.9%) Class II 34(25%) Class III 74(55.4%) Class VI 21(15.4%) |
| CHF | 71(52.2%) |
| Afib | Chronic 5 (3.7%) Acute 7(5.1%) |
| Obesity | 33(24.3%) |
| Smoking | 27(19.9%) |
| Dyslipidemic | 127(93.4%) |
| Diabetes Mellitus | 98(72.1%) |
| HTN | 79(58.1%) |
| Hypothyroidism | 12(8.8%) |
| PVD | 10(7.4%) |
| CVA | 10(7.4%) |
| COPD | 12(8.8%) |
| S.Creatinine | 136.9±125.6 (53-854) |
| Euro Score | 6.67±2.81(2-16) |
| Degree of MVR | +2 Moderate 17(12.5%) |
| +3 moderately severe 80(58.8%) | |
| +4 severe 39(28.7%) | |
| Tricuspid repair | Severe 22(16.2%) |
| LVEDD (mm) | 58.2± 6.9 [44-74mm] |
| LVESD (mm) | 44.4 ± 8 [25-63mm] |
| EF (%) | 38 ± 10.6 [18-60%] |
Table (2): Operative and postoperative data.
| Variable | Descriptive ( n=136) |
| Bypass time (min.) | 122.2±50.1 |
| Cross clamp time (min) | 101.8±43.5 |
| Prolonged ventilation >24hrs. | 35(25.7%) |
| Pneumonia or chest infection | 20(14.7%) |
| Sternal wound infection (SWI) |
25(18.4%) superficial SWI 2(1.5%) Deep SWI |
| Stroke | 3(2.2%) |
| Renal Failure | 8(5.9%) |
| Low cardiac output (LCO) | 42(30.9%) |
| Afib | 65(47.8%) |
| Grade I residual MR | 47 (34.6%) |
| Grade II residual MR | 16(11.8%) |
| Grade III residual MR | 3(2.2%) |
| Grade VI residual MR | 2(1.5%) |
| LVEDD (mm) | 54.8 ± 8.5 (33-75) |
| LVESD (mm) | 43.1 ± 9.3 (22-64) |
| EF (%) | 40.4 ± 10.9 (20-60) |
Very successful rate of repair without residual is (47.1%) and most postoperative residual MR is mild (34.6%).
Table (3): Postoperative death
| In hospital ≤ 30 days death | 3(2.2%) |
| Death during one year follow up | 3(2.2%) |
| Total mortality | 6 (4.4%) |
| Causes of death |
Cardiac 4 (2.9%) |
Table (4): Comparison between pre and postoperative echo data.
| Variable | Preoperative | Postoperative | P.value |
| LVEDD (mm) | 58.2 ± 6.9 | 54.8 ± 8.5 | <0.01** |
| LVESD (mm) | 44.4 ± 8.0 | 43.1 ± 9.3 | 0.05* |
| EF (%) | 38.2 ± 7.8 | 40.4 ± 10.9 | 0.01* |
LV dimensions are reduced postoperative and EF is improved significantly, when repair was performed by either band or ring.
Table (5): Mortality and other variables.
| Variable | Lived (n=130) | Died (n=6) | P.value (significant) |
| Preoperative LVEDD(mm) | 58.4 ± 6.8 | 52.7 ± 8.1 | 0.04* |
| Preoperative EF (%) | 38.4 ± 7.6 | 31.4 ± 6.1 | 0.03* |
| Postoperative EF (%) | 40.7 ± 10.8 | 22.5 ± 9.4 | 0.02* |
Table (6): Univariate and multivariate predictors of mortality.
| Variable | Univariate model Odds ratio P.value |
Multivariate model Odds ratio P.value |
| LVEDD(mm) | 1.02 (0.71-3.64)0.02* | 0.99 (0.64-3.28)0.04* |
| LVESD(mm) | 1.08 (0.68-3.57)<0.01** | 1.03 (0.65-3.51)<0.01** |
| EF (%) | 1.26 (0.87-4.29)<0.01** | 0.82-4.36)<0.01** 1.14 |
| Implant type ( ring) | Odd Ratio 6.16(0.34-112.1) |
P.value 0.22 |
Overall rate of mortality is (4.4%). MVr with low EF has a 3-fold increase in the risk of death compared with MVr with preserved EF (OR = 3.02, 95% CI 0.79-11.5, P = 0.03). Multivariable analysis showed that preoperative EF% (OR 1.14, 95%, CI 0.82–4.86, P = <0.01), preoperative LVESD (OR 1.03, 95% CI 0.65–3.51, P = <0.01) and preoperative LVEDD (OR 0.99, 95% CI 0.64–3.28, P = 0.04) were independent risk factors of mortality.
Discussion
Although both MVr and MVR may improve outcomes, repair has been associated with better short term outcomes and replacement with a more durable correction of MR.(5) We recommended MVr in IMR, if repairable, rather than CABG alone or MVR, with good results. MVr with a restrictive rigid ring has been associated with equal remodeling and mortality rates when compared with MVR. Although patients had higher MR recurrence following a repair, the reoperation rates did not differ between the repair and the replacement groups.(6)
IMR is considered to be a common cause of post infarction CHF and has been considered one of the therapeutic targets in heart failure. There is a relationship between the amount of non-viable LV segments, especially in posterior and inferior walls, and the recurrence of MR after the combined surgery.(7.8)
Combined MVr with CABG results in lesser postoperative MR compared with CABG alone. MVr was performed with downsized complete rigid annuloplasty ring which is the procedure of choice for IMR.(8,9) Complete annuloplasty ring is decreasing mitral leaflet strain and improving coaptation geometry, and enhances durability. Also, the Cosgrove flexible band is designed for preserving MA dynamics. It has been suggested that the saddle shape and the sphincter mechanism of the mitral valve can be preserved for 5- years after band implantation.(10)
Conclusion
CABG with MVr for non-mild IMR can be performed safely with improving symptoms, quality of life, decreased cost of readmission, low rate of MR recurrence, and without increasing mortality.
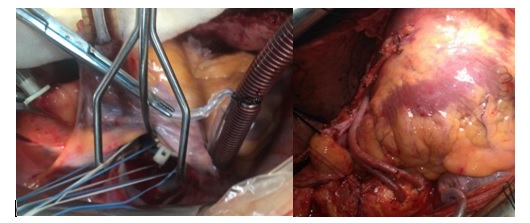
Figure (1):- Mitral Valve Repair with annuloplasty ring and CABG in cases of IMR
References
1. Varma PK, Krishna N, Jose RL and Madkaiker AN. Ischemic mitral regurgitation. Ann Cardiac Anesth 2017; 20: 432–439.
2. Malhotra A, Sharma P, Garg P, Bishnoi A, Kothari J and Pujara J. Ring annuloplasty for ischemic mitral regurgitation: a single center experience. Asian Cardiovasc Thorac Ann 2014; 22: 781–786.
3. LaPar D, Acker M, Gelijns A and Kron I. Repair orreplace for severe ischemic mitral regurgitation: prospective randomized multicenter data. Ann Cardiothorac Surg
2015; 4: 411–416.
4. Kulik A. Aortic root enlargement: worth the effort? Ann Thorac Surg 2010; 90: 703–705.
5. Ferket BS, Ailawadi G, Gelijns AC, et al. Cost-effectiveness of mitral valve repair versus replacement for severe ischemic mitral regurgitation. a randomized clinical trial from the Cardiothoracic Surgical Trials Network. CircCardiovascQual Outcomes 2018; 11: e004466.
6. Athanasopoulos LV, Casula RP, Punjabi PP, AbdullahiYS, Athanasiou T.A technical review of subvalvular techniques for repair of ischaemic mitral regurgitation and their associated echocardiographic and survival outcomes.Interact Cardiovasc Thorac Surg 2017; 25: 975–982.
7.Onorati F, Rubino AS, Marturano D, Pasceri E, Mascaro G, et al.Mid-term echocardiographic results with different rings following restrictive mitral annuloplasty for ischemic cardiomyopathy.EuroJ Cardiothorac Surg. 2009; 36: 250—260.
8.Sigita G, Egle U, Zaneta P, et al. Predictors of ischemic mitral regurgitation recurrence in patients undergoing combined surgery: additional value of cardiovascular magnetic resonance imaging. Interactive Cardiovasc Thorac Surg. 2018; 27: 222–28.
9. Mohammad H, Bahare S, Mohammad A, Farshid A, Farideh R . Long-term effect of papillary muscle approximation combined with ventriculoplasty on left ventricle function in patients with ischemic cardiomyopathy and functional mitral regurgitation. Euro J Cardiothorac Surg. 2011; 40: 756—60.
10. Wan S, Lee AP, Jin CN, et al.The choice of mitral annuloplastic ring—beyond “surgeon’s preference”. Ann Cardiothorac Surg. 2015; 4(3): 261-65.