Redefining Obesity Treatment: Integrating Pharmacotherapy and Surgery in the Modern Era

Obesity treatment is evolving beyond surgery. Emerging anti-obesity medications like semaglutide and tirzepatide offer significant weight loss, challenging bariatric surgery’s dominance. While surgery remains superior in long-term outcomes, an integrated, personalised approach combining pharmacotherapy and surgery may redefine care. Strategic synergy, not replacement, is key to optimal patient outcomes.
Introduction
Obesity is no longer a lifestyle issue—it is a chronic, relapsing, multifactorial disease with major global health consequences. It affects over 650 million adults worldwide, contributing to rising rates of type 2 diabetes, cardiovascular disease, non-alcoholic fatty liver disease (NAFLD), and various cancers. The mainstay of treatment traditionally revolved around behavioral modification, dietary changes, and exercise. However, these measures alone often prove inadequate due to biological resistance to weight loss.
For years, bariatric surgery has been the gold standard for sustained weight loss and resolution of comorbidities in individuals with severe obesity. Yet, the recent emergence of highly effective anti-obesity medications (AOMs) such as semaglutide (a GLP-1 receptor agonist) and tirzepatide (a dual GLP-1/GIP receptor agonist) has prompted a paradigm shift. These pharmacological options now offer substantial weight loss and metabolic improvement without invasive intervention. This raises an important clinical question: Will AOMs replace bariatric surgery, or are they best used in synergy?
Mechanisms of Action: Surgical and Pharmacological Pathways to Weight Loss
Bariatric Surgery encompasses procedures like sleeve gastrectomy (SG), Roux-en-Y gastric bypass (RYGB), and biliopancreatic diversion with duodenal switch (BPD-DS). These operations act via:
- Restriction of stomach volume,
- Malabsorption of nutrients (in some cases),
- Profound hormonal alterations including increased GLP-1, PYY, and reduced ghrelin,
- Resetting of hypothalamic satiety signaling and alteration of gut-brain axis dynamics.
Anti-Obesity Medications (AOMs), particularly incretin-based therapies, work pharmacologically by:
- Delaying gastric emptying,
- Enhancing insulin secretion (GLP-1 activity),
- Increasing satiety and reducing appetite through hypothalamic modulation,
- Promoting a negative energy balance while maintaining lean body mass to some extent.
Earlier medications like orlistat or phentermine-topiramate showed only modest effects and were often limited by side effects. The new generation—especially semaglutide (Wegovy) and tirzepatide (Zepbound)—has changed the narrative by mimicking the hormonal alterations seen after bariatric surgery.
Comparing Weight Loss Outcomes
Weight loss effectiveness is a central metric in comparing AOMs and bariatric surgery.
| Intervention | % Total Body Weight Loss (TBWL) | Time Frame | Durability |
| Semaglutide (2.4 mg) | ~15% | 68 weeks (STEP trials | Requires continuation |
| Tirzepatide (15 mg) | ~20–22% | 72 weeks (SURMOUNT-1) | Requires continuation |
| Sleeve Gastrectomy | 25–30% | 1–2 years | 10+ years |
| RYGB | 30–35% | 1–2 years | 10+ years |
| BPD-DS | 35–45% | 2 years | Longest duration |
While tirzepatide approaches the efficacy of surgery, most pharmacological weight loss depends on indefinite continuation. Once the medication is stopped, a significant portion of weight is typically regained, as demonstrated in the STEP-4 trial, where patients regained 2/3 of their lost weight within a year of stopping semaglutide.
In contrast, bariatric surgery achieves metabolic reprogramming that persists even after weight plateaus, although some degree of long-term weight regain is still common.
Impact on Comorbidities
A key argument for surgery lies not only in weight loss but in the remission or resolution of obesity-related diseases.
- Type 2 Diabetes (T2D):
Bariatric surgery induces remission in up to 80% of patients in the first few years post-op. The STAMPEDE trial showed that RYGB and SG significantly outperformed medical therapy in glycemic control. AOMs like semaglutide and tirzepatide also reduce HbA1c by 1.5–2.5%, but they rarely induce remission unless combined with significant lifestyle change. - Cardiovascular Risk:
Surgery improves lipid profiles, reduces hypertension, and lowers cardiovascular mortality by up to 40% according to the Swedish Obese Subjects (SOS) study. Recent trials (e.g., SELECT) are beginning to show that semaglutide also reduces major adverse cardiovascular events (MACE) in high-risk patients with obesity, though long-term mortality data are still pending. - Fatty Liver Disease:
NAFLD and NASH improve dramatically after RYGB and SG due to insulin sensitivity changes. AOMs are now also under investigation for NASH resolution (e.g., semaglutide, resmetirom), with promising results in fibrosis reduction. - Sleep Apnea, PCOS, GERD:
These conditions tend to improve more consistently with surgery, particularly where mechanical relief (e.g., reduction in abdominal pressure) is necessary. AOMs are still being evaluated for their role in these syndromes.
Safety Profiles: Weighing the Risks
| Metric | Bariatric Surgery | Anti-Obesity Medications |
| Acute Risks | Surgical site infections, leaks, bleeding, VTE | Nausea, vomiting, pancreatitis (rare), gallstones |
| Long-term Risks | Nutritional deficiencies, internal hernias, marginal ulcers | Weight regain after discontinuation, GI intolerance |
| Mortality | 0.1–0.5% in experienced centers | Extremely low in clinical trials |
| Monitoring Requirements | Lifelong micronutrient follow-up | Periodic labs, monitoring for pancreatitis or thyroid issues |
Despite concerns, bariatric surgery has become remarkably safe, with complication rates lower than gallbladder surgery in many centers. Meanwhile, AOMs offer a non-invasive alternative but carry the burden of indefinite use, unknown long-term safety (beyond 2–3 years), and limited real-world adherence.
Cost and Access: A Global Disparity
| Category | Bariatric Surgery | AOMs (e.g., semaglutide, tirzepatide) |
| Initial Cost | $10,000–30,000 (surgery + hospital stay) | $12,000–16,000 per year (uninsured) |
| Insurance Coverage | Covered in many countries with strict criteria | Poor or no coverage in most regions |
| Cost-Effectiveness | Proven over 10–15 years due to reduction in comorbidities | Debated; high dropout and weight regain rate |
Cost and access are major limiting factors. In many middle-income countries, surgery is inaccessible for economic reasons, and AOMs are not covered by insurance. For systems like the NHS or Medicare, widespread AOM use could carry significant budget implications if not shown to reduce hard endpoints like mortality.
Patient Preference and Adherence
Surveys show that many patients prefer non-invasive therapies when available, even if the weight loss is less durable. This is particularly true for:
- Younger individuals,
- Those afraid of surgery,
- Patients with lower BMIs,
- Individuals with previous poor surgical experiences.
However, real-world adherence to AOMs drops sharply beyond 12 months. Side effects, out-of-pocket costs, and the psychological impact of limited progress can drive discontinuation. Conversely, surgery offers a more binary commitment, with irreversible anatomical changes forcing behavior modification.
Synergistic Potential: Not Mutually Exclusive
Rather than viewing AOMs and surgery as mutually exclusive, a hybrid model is gaining traction.
- Preoperative AOM Use: Short-term AOM use before surgery may reduce liver volume, operative time, and anesthesia risk in high-BMI patients.
- Postoperative AOM Use: For patients who plateau or regain weight after surgery, GLP-1-based therapy may help reinitiate weight loss.
- Combination Therapy: Studies are exploring combining endoscopic therapies with AOMs, or even AOMs + surgery for super-obese individuals (BMI >60 kg/m²).
This integrated model could allow physicians to personalise treatment sequences and optimise patient outcomes.
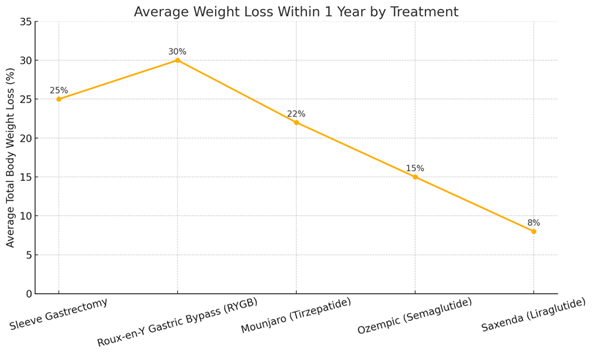
Figure 1: Average Weight Loss within One Year by Treatment
| Intervention | % Total Body Weight Loss (TBWL) | Time Frame | Durability |
| Sleeve Gastrectomy | ~25% | 12 months | Durable >10 years |
| RYGB | ~30% | 12 months | Durable >10 years |
| Mounjaro (Tirzepatide) | ~22% | 72 weeks | Requires continuation |
| Ozempic (Semaglutide) | ~15% | 68 weeks | Requires continuation |
| Saxenda (Liraglutide) | ~8% | 12 months | Requires continuation |
Interpretation:
While sleeve gastrectomy and RYGB provide the most significant and durable weight loss, Mounjaro (tirzepatide) shows remarkably strong performance among pharmacologic agents, nearing surgical efficacy. Semaglutide and liraglutide offer moderate reductions but still fall short of surgical benchmarks. The need for continuous medication use in AOMs highlights the dependency on adherence and long-term tolerability.
Future Outlook: Personalised, Tiered Treatment Algorithms
The future of obesity management lies in personalised medicine. Predictive markers—genomic, metabolic, behavioral—may help stratify patients based on likely response to surgery vs AOMs.
- Who benefits most from surgery?
Those with long-standing T2D, mechanical complications (e.g., sleep apnea, GERD), and need for durable, life-altering change. - Who benefits most from AOMs?
Early-stage obesity, younger adults, or patients with contraindications to surgery.
In parallel, next-generation agents—including triple agonists (e.g., GLP-1/GIP/glucagon), long-acting oral formulations, and combination regimens—may close the efficacy gap even further.
Conclusion
Bariatric surgery remains unmatched in long-term weight loss and remission of obesity-related comorbidities, particularly for those with severe or complicated obesity. Yet, the landscape has shifted. New anti-obesity medications offer potent, flexible, and increasingly safe options for a broader range of patients—many of whom might never have considered surgery.
Rather than viewing the two as competitors, the most forward-thinking approach is one of integration and individualisation. The right treatment, at the right time, for the right patient will ultimately lead to the best outcomes. As science continues to evolve, clinicians must embrace both tools in the arsenal against one of the 21st century’s greatest health threats.