Precision vs Price: Navigating the Therapeutic Dilemma in India's Battle with NCDs
Role of precision medicine and pharmacogenomics in addressing non-communicable diseases in India is persistently growing, where integration of polygenic risk scores, national genomic initiatives into healthcare is needed at the time. Emphasis is placed on challenges, opportunities, and policy support needed to advance personalised, cost-effective treatment strategies.
Non-communicable disease
India, the second most populous country globally, exhibits significant genetic heterogeneity due to diverse cultural, social, and biological behaviour. According to the 2017 report “India: Health of the Nation's States” by the Indian Council of Medical Research (ICMR), the share of deaths attributed to Non-Communicable Diseases (NCDs) in India rose sharply from 37.9% in 1990 to 61.8% in 2016.1 The four major NCDs— diabetes, cancers, cardiovascular diseases, and chronic respiratory diseases—pose a growing public health burden. These conditions are primarily driven by common behavioural risk factors: physical inactivity, unhealthy diets and the use of tobacco and alcohol.1,2 The Economic Survey of India 2023–24, presented by the Union Finance Minister on July 22, 2024, during the opening day of the Budget Session, emphasised that enhancing the health status of India’s youth is of vital importance. Obesity is a major concern in youth due to an unhealthy diet.
The preventive aspect of NCDs is being reinforced through the Ayushman Bharat Health and Wellness Centre scheme, NPCDCS, which promotes wellness activities and targeted health communication at the community level. As part of this initiative, individuals over 30 years of age are screened for common NCDs, including breast and cervical cancer screening among women. Public awareness efforts include observing national and international health days, utilising various media platforms, and promoting healthy eating through the Food Safety and Standards Authority of India (FSSAI). Additionally, initiatives for a healthy lifestyle, the Fit India Movement by the Ministry of Youth Affairs and Sports and Yoga programs by the Ministry of AYUSH.1
Precision medicine (PM)
PM is a form of medicine that uses information about a person’s genes, proteins, environment, and lifestyle to prevent, diagnose, or treat disease.3 Also known as “personalised medicine”. With rapid advancements in genomics, pharmacogenetics has evolved from studying single gene–drug interactions to the more comprehensive field of pharmacogenomics. This shift now integrates insights from various "-omics" disciplines such as proteomics, transcriptomics, metabolomics, and metagenomics, enhancing the precision and effectiveness of medical interventions.3
History
In 1909, Archibald Garrod introduced the concept that inborn errors of metabolism could underlie certain diseases and noted that drug responses vary significantly among individuals—what is safe for most may be toxic for some, while others may show unusual tolerance. The role of genetics in drug response was further highlighted by Arthur L. The term “pharmacogenetics” was coined by Friedrich Vogel in 1959 following discoveries of inherited enzyme deficiencies, such as in N-acetyl transferase, glucose-6-phosphate dehydrogenase (G6PD) and plasma cholinesterase. These findings, along with the identification of CYP2D6 deficiency linked to adverse reactions to drugs like debrisoquin and sparteine, established the genetic basis of variability in response to xenobiotics.3
Transition to Pharmacogenomics
Sequencing the human genome in 2001 has led to rapid advances in genomic sciences, focusing our attention not just on single genes but on the ensemble of all genes and genomic functions. This broader perspective has given rise to the term “pharmacogenomics,” which, as defined by the National Human Genome Research Institute, focuses on using DNA and amino acid sequence data to guide drug development and testing, though many reviews still primarily reflect pharmacogenetic concepts centred on the influence of one or a few genes on drug response.3
Emerging therapeutic approaches extending beyond traditional drugs are significantly expanding the scope of pharmacogenomics. These innovative modalities include RNA and DNA molecules, gene editing and gene therapy techniques, biologics like proteins, live cells, tissues, beneficial microbes, and viral vectors. Additionally, complementary strategies such as radiation, nutritional interventions, and physical activity are increasingly integrated into personalised treatment plans.3
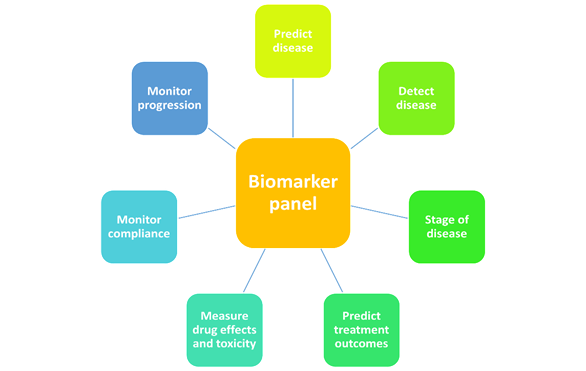
The use of biomarkers in personalised medicine involves both phenotypic and genomic indicators, with germline mutations detectable at any time, while other markers—such as somatic mutations—can fluctuate depending on disease progression, age, treatment, and various external factors.3,4 (Figure 1)
Figure 1: Use of biomarkers in personalised medicine
Genome data status in India
India’s efforts in genomic research began as early as 2003 with the launch of the Indian Genome Variation database project, which aimed to document both known and novel single-nucleotide polymorphisms (SNPs) and repeats across over 1,000 genes in 15,000 individuals from diverse regions of the country. A major milestone followed with the launch of the “Genome India Project” on January 3, 2020, by the Department of Biotechnology, in collaboration with 20 research institutes, which seeks to collect genetic data from 10,000 individuals nationwide to develop a comprehensive Indian reference genome. Through whole-genome sequencing, this initiative aims to create an extensive catalogue of genetic variation within Indian population, paving the way for advancements in next-generation precision medicine. Additionally, innovative research areas are emerging, such as Ayurgenomics—a fusion of Ayurveda and genomics for personalized care—and the Indian Cancer Genome Atlas, developed in collaboration with The Cancer Genome Atlas and the Department of Biotechnology, to build a detailed omics database of Indian cancer patients.5 Including, the Paediatric Rare Genetic Disorders (PRaGeD) mission aims to explore novel genes and genetic variants, provide child-specific treatment strategies for rare inherited conditions.6
Polygenic risk scores (PRS)
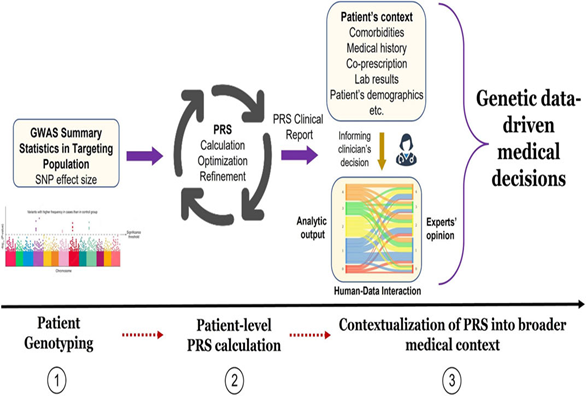
Polygenic risk scores (PRS) are the emerging tool in pharmacogenomics that estimate a patient-level genetic predisposition to a specific trait or disease. PRS calculate the effects of multiple genome-wide risk variants, weighted by their impact as identified through GWAS (Genome-Wide Association Study) data, though it does not account for gene-gene or gene-environment interactions. By capturing the cumulative effect of many small genetic variations, PRS single numeric index, making it an efficient and convenient method for predicting complex disease risk and enabling large-scale population screening.7 The various steps involved in calculating PRS and implementation of PRS in clinical decision making for disease involve patient genotyping, patient-level PRS calculation and contextualization of PRS into a broader medical context.8,9 (Figures 2 & 3)
Figure 2: Steps involved in polygenic risk score (PRS) calculation9
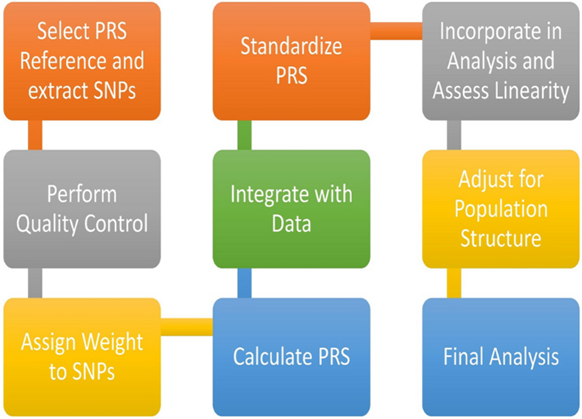
Figure 3. Implementation of PRS results into medical decision process8
Polygenic risk scores in disease risk prediction
Pemmasani et al. concluded that the polygenic risk score (PRS) model specifically developed for the Indian population by using imputed genotype data from UK Biobank and testing the developed PRS on data from GenomegaDB of Indians living in India, demonstrated strong accuracy in predicting the likelihood of developing type 2 diabetes (T2D).4
Genome-wide polygenic risk scores for five common diseases revealed that 8.0%, 6.1%, 3.5%, 3.2%, and 1.5% of the population are at more than three times the normal risk for coronary artery disease, atrial fibrillation, type 2 diabetes, inflammatory bowel disease, and breast cancer, respectively. Notably, for coronary artery disease, this high-risk group is about 20 times more common than individuals carrying rare monogenic mutations that confer a similar level of risk.10
Cross et al. have reviewed commonly used medications examined in PRS studies for predicting adverse drug reactions (ADRs) and treatment efficacy. Widely prescribed drugs such as statins, clopidogrel, warfarin (increasingly replaced by direct oral anticoagulants), antipsychotics, and antidepressants have shown varying degrees of genetic association with treatment outcomes. However, the overall contribution of polygenic risk scores in explaining phenotype variability remains limited. This underscores the need for continued research and refinement in pharmacogenomic studies to enhance predictive accuracy and clinical utility.11
In predicting osteoporotic fractures, PRS use reduced the number of patients requiring screening by 41% while maintaining high sensitivity and specificity. This highlights the potential of PRS not only for identifying high-risk individuals but also for effectively ruling out those unlikely to develop complex, low-prevalence conditions, thereby improving predictive precision.8
Challenges
Globally, evidence supports the therapeutic and economic benefits of pharmacogenomics, but also not surprising that using PRS in pharmacogenomics and modelling of pharmacoeconomic for pharmacogenomic are not easily overcome.
- Regulatory law for data management and maintenance of BioBanks is deficient, which may hamper the trust of participants.6
- One of the key challenges in pharmacogenomic research is the need for high-fidelity data on patients’ drug response phenotypes. To ensure meaningful comparisons, studies must include uniformly treated patients and use clearly defined clinical endpoints—such as standardised scales for disorders like depression—captured within a precise time frame. Such detailed and granular data is typically available only in well-structured, patient-level clinical databases, limiting the scope and accessibility of research.8
- Polypharmacy—the concurrent use of multiple medications (typically more than five)—raises the risk of drug-drug interactions. Additionally, underlying conditions such as kidney failure can heighten susceptibility to drug toxicity. Both factors can complicate the interpretation of genetic influences by altering the apparent effect sizes in pharmacogenomic studies.8
- With the rapid decline in the cost of genetic testing, traditional pharmacoeconomic models may no longer accurately represent the evolving healthcare landscape.
- There is a lack of clear guidelines for evaluating pharmacogenomic evidence. This makes it difficult to assess its clinical utility. As a result, broader adoption of such testing is often delayed.
- Clinician unfamiliarity with billing logistics: Many clinicians may face difficulties navigating the billing process for pharmacogenomic testing, which can be a barrier to implementation.12
Future of PRS and Pharmacogenomics in India
Two critical aspects of evidence-based clinical interventions are treatment costs and the protection of patient rights. Integrating pharmacogenomic information into electronic health records, along with built-in clinical decision support tools, will be crucial for advancing and scaling personalized treatment strategies. The diverse range of genetic, clinical, and environmental factors—such as sex, age, socioeconomic conditions, social determinants of health, body weight, and nutritional status—requires large-scale data integration. Leveraging artificial intelligence will be essential to synthesize these complex variables and provide clinically meaningful insights for guiding personalised medicine.3
Despite existing challenges, the integration of Pharmacoeconomics and pharmacogenomics offers significant potential for improving healthcare outcomes. With declining costs of genetic testing and growing evidence of its clinical and economic value, strategies like pre-emptive pharmacogenomic testing are proving effective in reducing adverse drug reactions and enhancing treatment success. Moving forward, collaboration among healthcare providers, policymakers, and payers will be essential to establish standardised guidelines, ensure consistent evaluation, and promote equitable access to pharmacogenomic services.12
India’s personalised medicine sector is expanding quickly, with genetic testing—an important part of this field—experiencing strong growth. In 2022, the Indian genetic testing market was valued at about ₹4,544.5 crore (roughly $61 million) and is expected to grow even further by 2028. This rapid growth highlights the growing importance of personalised medicine in shaping the future of healthcare in India.13
Many highly differentiated SNPs, indels, and haplotypes remain unverified for their association with drug response or toxicity due to limited evidence. Implementing dosing guidelines tailored to genetic variations prevalent in the Indian population can actively improve treatment outcomes and patient safety 14
Global scenario
Countries such as the U.K., U.S., Japan, China, and various European nations have established legal frameworks or detailed regulatory guidelines that govern key aspects of biobanking. These include provisions for obtaining informed consent, ensuring participants' right to withdraw, safeguarding privacy, and protecting personal data.6
Conclusion
Integrating precision medicine into medical education, collaboration among clinicians, pharmacologists, bioinformaticians, programmers, biostatisticians, and other experts. To ensure its effective implementation, innovations in data storage, point-of-care access by integrating artificial intelligence, and a clear long-term government policy roadmap will be impactful in navigating the dilemma of prevention and treatment of non-communicable diseases in India.
References
- Status of Non-Communicable Diseases (NCDs) in India. PIB Delhi; 2022 Feb p. 1. Report No.: Release ID: 1796435.
- Anjana RM, Unnikrishnan R, Deepa M, Pradeepa R, Tandon N, Das AK, et al. Metabolic non-communicable disease health report of India: the ICMR-INDIAB national cross-sectional study (ICMR-INDIAB-17). Lancet Diabetes Endocrinol. 2023 Jul;11(7):474–89.
- Sadee W, Wang D, Hartmann K, Toland AE. Pharmacogenomics: Driving Personalized Medicine. Pharmacol Rev. 2023 Jul;75(4):789–814.
- Pemmasani SK, Atmakuri S, Acharya A. Genome-wide polygenic risk score for type 2 diabetes in Indian population. Sci Rep. 2023 Jul 18;13(1):11568.
- Yadav A, Sagar R. Precision medicine in public health in India: Nascent but poised in the right direction. Indian J Public Health. 2021;65(4):414.
- Precision Medicine in India - Current Affairs [Internet]. [cited 2025 Jun 27]. Available from: https://www.nextias.com/ca/current-affairs/14-10-2024/precision-medicine-in-india
- Choi SW, Mak TSH, O’Reilly PF. Tutorial: a guide to performing polygenic risk score analyses. Nat Protoc. 2020 Sep 1;15(9):2759–72.
- Simona A, Song W, Bates DW, Samer CF. Polygenic risk scores in pharmacogenomics: opportunities and challenges—a mini review. Front Genet. 2023 Jun 15;14:1217049.
- Singh O, Verma M, Dahiya N, Senapati S, Kakkar R, Kalra S. Integrating Polygenic Risk Scores (PRS) for Personalized Diabetes Care: Advancing Clinical Practice with Tailored Pharmacological Approaches. Diabetes Ther. 2025 Feb;16(2):149–68.
- Khera AV, Chaffin M, Aragam KG, Haas ME, Roselli C, Choi SH, et al. Genome-wide polygenic scores for common diseases identify individuals with risk equivalent to monogenic mutations. Nat Genet. 2018 Sep;50(9):1219–24.
- Cross B, Turner R, Pirmohamed M. Polygenic risk scores: An overview from bench to bedside for personalised medicine. Front Genet. 2022 Nov 11;13:1000667.
- The Evolving Landscape of Pharmacoeconomics in the Era of Pharmacogenomics [Internet]. Pharmacoeconomics. 2024. Available from: https://dromicslabs.com/
- More S. What Is The Impact Of Personalised Medicine On Indian Healthcare? [Internet]. What Is The Impact Of Personalised Medicine On Indian Healthcare? 2025. Available from: https://www.mrmed.in/health-library/health-care/personalized-medicine-india?srsltid=AfmBOoqFN_z0KRi85eak9QyI62lFGStURnL2rqQkVYHo3DWWFVF3V_zt
- Sahana S, Bhoyar RC, Sivadas A, Jain A, Imran M, Rophina M, et al. Pharmacogenomic landscape of Indian population using whole genomes. Clin Transl Sci. 2022 Apr;15(4):866–77.









-(1).jpg)
