Introduction
There is no doubt that advances in the performance of diagnostic imaging devices have improved the quality of medical diagnoses. Due to technological improvements, the image quality has dramatically increased, such as the adoption of higher frequency and broadband probes in Ultrasonography (US), the development of stronger magnetic fields in Magnetic Resonance Imaging (MRI), and the incorporation of multi-row detectors in Computed Tomography (CT). In addition, to these improvements for such device technologies, the environment of diagnostic imaging has also changed due to the development of medical image information processing. However, in recent decades, no new diagnostic imaging devices have been developed.
Kyoto University Hospital has researched and developed next-generation image diagnostic technologies in collaboration with Canon Inc., which was named the ‘Innovative Techno-Hub for Integrated Medical Bio-imaging’ (CK Project), during 2006-2015. As a result of our efforts to develop technologies related to Photoacoustic (PA) diagnostic equipment, we showed that this approach can be applied to diagnostic devices based on new principles. In that time, we demonstrated the diagnostic performance for breast cancer patients by exploratory clinical research. Afterwards, the project was taken over by the Impulsing Paradigm Change through Disruptive Technologies Program (ImPACT) led by the Council for Science, Technology and Innovation (CSTI) of the Cabinet Office, Japan, and here, we are promoting research and development. Our version of ImPACT, called the ImPACT Yagi programme [1] (named after of our programme manager), promotes the development of component technology and its practical application as well as clinical research for diagnosis targeted by PA technology. In this article, we present an overview of PA imaging, introduce examples of our results, and discuss the direction of future medical use and healthcare applications.
Principles of PA imaging
The PA effect is a phenomenon in which ultrasonic waves are generated by irradiating light on a light-absorbing substance. In the case of a living body, pulsed light is irradiated onto the surface of the body and is transmitted through the body, where light energy is absorbed by “absorbers” such as haemoglobin. Rapid and instantaneous thermal expansion occurs, and energy is recovered at the absorber by pulsed light energy, finally generating ultrasonic waves. From the PA signals detected at multiple positions outside the body, image reconstruction is performed as an initial acoustic pressure image. The ultrasonic waves transmitted through the body with less scattering than light, allowing high-resolution haemoglobin images (i.e. blood vessel imaging) to be obtained. If we can calculate the light fluency in the living body, the absorption coefficient distribution can be imaged. This imaging method is called PAT.
The PA effect itself was first reported in 1881 [2], but the research aiming at its application to medical imaging began in the 1990s [3], most likely because the application to living organisms was realised by the digitisation of ultrasonic signal processing and high-speed image reconstruction technology was achieved by the increased processing speed of microprocessors. PAT imaging has an imaging depth of several centimetres and a spatial resolution corresponding to that of ultrasonic imaging. PAT also has the unique ability for optical imaging, as occurs in an image reflecting the light absorption characteristics of an object. By using a plurality of near infrared wavelength light matching the light absorption coefficient of deoxygenated haemoglobin and oxygenated haemoglobin, one can image the absorption coefficient distribution and oxygen saturation distribution of the blood in the living body with high resolution. To clinically apply this technique, we first focused on applying PA imaging to breast cancer diagnosis, the focus of our research [4, 5]. To solve the technical problems revealed in this research, we changed the configuration of the breast holding method from the sandwich method between parallel flat plates using a flat shape probe to the cup method with a Hemispherical Detector Array (HDA) system [6] in the ImPACT Yagi programme (Figure 1). An HDA is automatically scanned after simply immersing an object (such as a breast or a hand) in a water cup for acoustic matching, and a PA image of the subject can be automatically obtained over a wide range [7, 8]. Figure 2 presents a blood vessel image of the palm. We can obtain the fine vascular structures in humans with non-invasive and non-ionising radiation, which is difficult to achieve with conventional modalities.
Clinical research candidate
PAT can non-invasively image the vascular network and the blood state with high resolution under the skin of a living body, offering new possibilities for clinical applications.
Figure 3 illustrates the experimental setup of our PAT system, and Figures 4 (a)-(c) show an example of a PAT image obtained by our PAT apparatus with an HDA. The breast shape in MRI scanning was deformed to that in PAT scanning in the digital data by using the thin plate spline technique, and these two images are overlaid. As is clear in Figure 4(c), the tumour-related blood vessels run towards the central part of the tumour [6]. Since, PAT is non-invasive and uses non-ionising radiation, it can be used frequently. For example, PAT imaging can be used to monitor and evaluate the effects of drug treatment and to individualise drug therapy.
We are exploring the application of this technology for the diagnosis of other diseases. We consider not only diseases appearing on the skin’s surface but also major diseases such as arteriosclerosis, cancer and rheumatism diagnosed candidates for our research. Although, it is known that the symptoms and conditions of these diseases are expressed in blood vessels and circulatory states, MRI and CT are invasive and cannot be used frequently due to contrast media and radiation exposure. Ultrasonic methods are simple and non-invasive, but they are dependent on the examiners' skills and are inferior in terms of reproducibility. For these reasons, evaluations of blood vessels and circulation status have not been applied to the prevention and early diagnosis methods. If we can image the vascular network and blood condition with non-invasive and non-ionising radiation using PAT, we can expect early detection and disease diagnosis in many diseases using blood vessel images as imaging biomarkers. Furthermore, if assessments such as the normalisation of blood vessels and skin ageing are developed, it will be possible to evaluate therapeutic effects and to develop cosmetic and healthcare monitoring.
We set up a newly completed wide-field visualisation system and we will develop a new diagnostic method of vascular imaging in several departments and demonstrate its effectiveness. Furthermore, we will analyse image information and medical information to realise pre-emptive medical care and to construct a health risk prediction model. We will introduce the same wide-field visualisation system to Keio University Hospital, where we intend to promote clinical research while cooperating between the two universities.
Summary
PA imaging is a novel technique that can visualise vascular structures and oxygen saturation status, details that cannot be seen by conventional modalities. In addition, PA imaging can be used frequently to examine the effects of drug treatment on a patient and be easily used to diagnose healthy subjects because it is a non-invasive vascular imaging technique that uses non-ionising radiation. Furthermore, PA imaging is expected to contribute to ultra-early diagnosis, assessment of physical function deterioration and pre-emptive medical care by adopting it for medical checkups. We hope to improve diagnostic methods of PAT through clinical research by improving the device analytical technology and contribute to vascular health.
References
[1]http://www.jst.go.jp/impact/hp_yagi/index.html
[2]Bell AG: Production of Sound by Radiant Energy, Popular Science Monthly. 1881;19:186-197
[3]Kruger RA, et al.: Photoacoustic ultrasound: theory, Proc SPIE. 1994; 2134A:114-8.
[4]Kitai T, et al.: Photoacoustic mammography: initial clinical results, Breast Cancer. 2014 Mar;21(2):146-53.
[5]Asao Y, et al.: Photoacoustic mammography capable of simultaneously acquiring photoacoustic and ultrasound images. J.Biomed. Opt., 21(11), 116009, doi: 10.1117/1.JBO.21.11.116009 (2016).
[6]Toi M et al.: Visualization of tumor-related blood vessels in human breast by photo acoustic imaging system with a hemispherical detector array. Sci.Reports, 7:41970, DOI:10.1038/srep41970 (2017)
[7]THE CANON FRONTIER 2017/2018;17, http://www.canon.com/technology/pdf/canon-frontier-2017-2018-e.pdf
[8]Matsumoto Y et al.: Label-free photoacoustic imaging of human palmar vessels: a structural morphological analysis. Sci.Reports, 8:786, DOI:10.1038/s41598-018-19161-z (2018)
Acknowledgements
This work was funded by the ImPACT of the Council for Science, Technology and Innovation (Cabinet Office, Government of Japan).
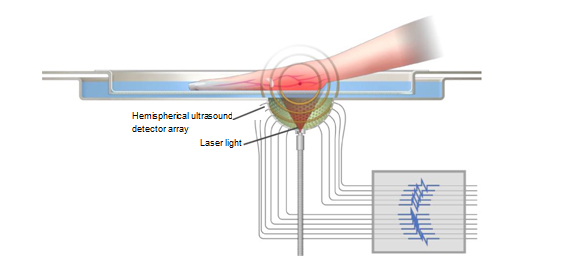
Fig. 1 Device configuration for PA imaging with a hemispherical ultrasound detector array.
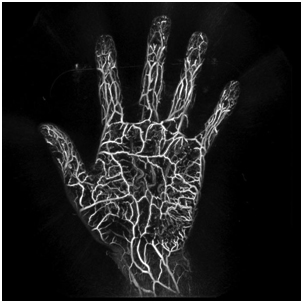
Fig. 2 Example of a blood vessel image of the palm taken at a wavelength of 795 nm using our PA imaging system.

Fig. 3 PAT system designed for breast cancer diagnosis [4]. To image the breast, the patient lies prone on the bed and places one breast into the spherically shaped cup indicated by the yellow arrow.
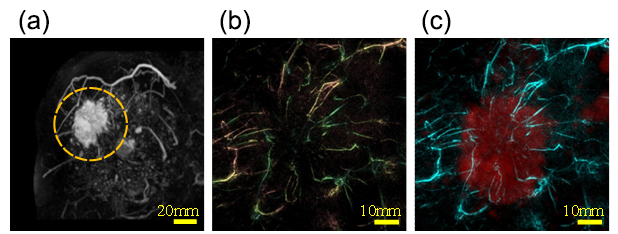
Fig. 4 Images obtained with MRI and PAT [6]. (a) MRI image (the yellow broken line represents a tumour lesion), (b) PAT image, (c) MRI image (red) and PAT image (cyan). The MRI image was overlaid onto the PAT image after the MRI image was deformed into a breast shape of the PAT image by image processing technology.