This article recommends a novel surgical technique for the treatment of complete atrioventricular septal defect (CAVSD): V-shaped double-layer patch technique. Since 2011, V-shaped double-layer patch technique applied for all CAVSD in our centre which had a satisfactory survival rate and reoperation-free rate during follow-up.
Currently popular surgical techniques for complete atrioventricular septal defect (CAVSD) are mainly ‘two-patch technique’ and ‘modified single-patch technique’. The discussion on which surgical strategy to use (two-patch versus modified single-patch technique) has continued for more than 20 years. As pointed out by Prof. Carl L. Backer, an internationally renowned authoritative expert in congenital heart surgery, cardiac surgeons have always been troubled by reoperation due to postoperative left atrioventricular valve regurgitation. Here in our centre, we applied a novel surgical technique ‘V-shaped double-layer patch technique’. It is a functional surgical technique, in which the left atrioventricular valve (LAVV) was protected by maintaining its original shape and position, avoiding being pulled by the patch tension. The follow-up (the longest 10 years, and the median 5.1 years) results have exhibited excellent performance of this technique.
The surgical technique for CAVSD should be designed to have anatomic advantages and to be more consistent with the principle of haemodynamic, so as to reduce the postoperative complications and improve prognosis. The V-shaped double-layer patch technique procedures proved to reduce the reoperation rate. Moreover, it is simple and takes no more operating time than other techniques.
We noticed some something interesting: (1) severe AV regurgitation and LV outflow tract stenosis were less common in partial atrioventricular septal defect (PAVSD), and the long-term prognosis was much better than CAVSD. (2) Some CAVSD cases had no or only mild AV regurgitation before operation, and the common atrioventricular valve (AV) regurgitation may be characterised by progressive aggravation. (3) We consecutively observed 111 cases of fetal CAVSD echocardiography and found that 94 per cent had no or mild AV valve regurgitation (56 cases without and 48 cases with mild), and only 1 case (0.9 per cent) had severe AV valve regurgitation. Therefore, we consider that in spite of the congenital defects of the AV valve and subvalvular structure of CAVSD, most of them still have a certain haemodynamic structural rationality, and their insufficiency is the result of long-term volume overload. Therefore, it is critical to maintain the original shape and position of the AV and its subvalvular structures for CAVSD patients.
Both the classical single-patch and two-patch techniques change the shape and the position of the AV and subvalvular structures. Due to the higher pressure in the left heart than in the right, the patch separating the left and the right created a continuous tension to the right ventricle, and the AV sutured on the patch would transmit the tension to the LAVV, making the patch concave on the left side, which may lead to the possibility of dehiscence at that suture line and the LAVV regurgitation.
The operation of CAVSD has two purposes: (1) to repair the atrial and ventricular septal defect and prevent blood shunting from left to right; (2) to repair and restore the function of the AV valve, which is the key to the success of the operation and affects the long-term outcome. We think that the haemodynamics of the AV valve is similar to the aerodynamics of a parachute, and that changing the original state of the AV valve and subvalvular structure is as bad as changing the parachute's canopy shape and cord length. If the ventricular septal defect under the AV valve is regarded as defect of the root part of the LAVV, the left patch can also compensate for the loss of suture and increase the area of the AV valve. Then the AV valve and the subvalvular structure can maintain their original shape and position and keep their function of opening and closing unchanged as before the operation. The right patch is sutured on the anterior portion of interventricular defect and the edge of the atrial septal defect. It reduces the repaired margin of ventricular septal defect by 50 per cent and decreased the incidence of ventricular septal defect residual. The right ventricle is a low-pressure heart chamber, and the right AV valve is under less pressure. The right AV valve is fixed in the proper position of the patch to achieve the purpose of repairing the right AV valve. In other words, the "V-shaped doublelayer patch technique" is formed by the patch for repairing the root of the LAVV and the patch for repairing the atrial and ventricular septal defect (Figure 1).
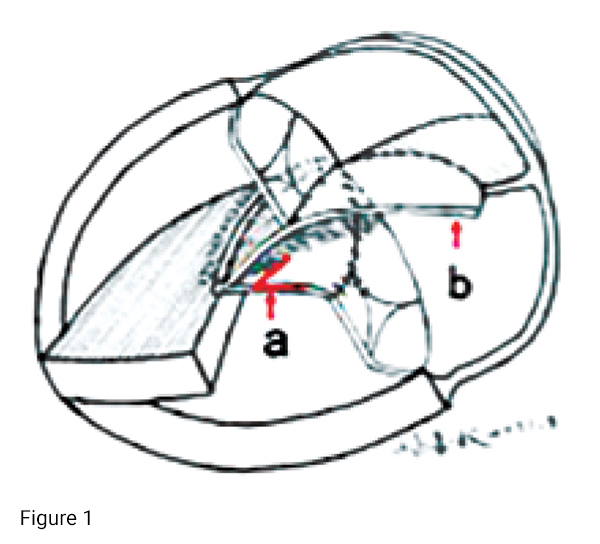
The operation is performed by conventional median sternotomy under continuous CPB through aortic and bicaval cannulae with cooling to 25-28°C. Cold histidine-tryptophanketoglutarate cardioplegia solution (Custodiol; Dr F Kohler Chemie GmbH, Alsbach, Germany) was given for myocardial protection.
1. Extracting and folding method of untreated autologous pericardium patch: Remove the fibrous tissue on the autologous pericardium. Keep the smooth surface of the pericardium inward, sew 6 stitches for traction and the folding of the patch is completed. The length of the patch is a plus b, and the width of the patch is c (Figure 2). When extracting the autologous pericardium, its length and width should be 20 per cent larger than the measured value, so that it can be trimmed to an appropriate size for suture.
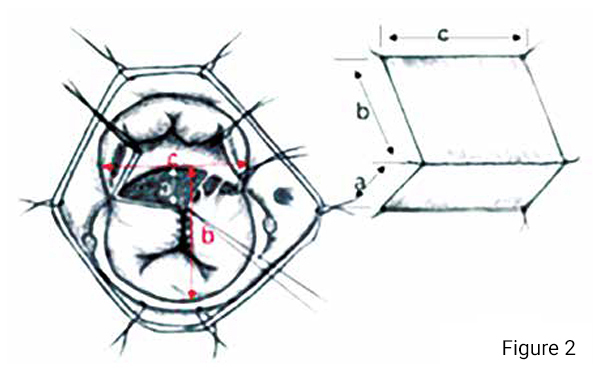
2. Observe the depth and the deepest position of the ventricular septal defect under the AV valve. Observe the edges of the left superior leaflet (LSL) and the left inferior leaflet (LIL) during saline injection. Suture interruptedly with the first needle for traction (Figure 2).
3. For Rastelli type-A and type–B CAVSD patients, the incision of the bridging leaflets is not necessary. Only for Rastelli type-C and very few type-B patients, the incision is required. The bridging leaflets across the interventricular septal crest was cut at the corresponding position to facilitate the placement of the patch (Figure 2). When the anterior bridge valve and the posterior bridge valve are incised, pay attention that the incision should be in the middle of the boundary between the left and right ventricular chordae tendineae to avoid damage to the subvalvular structure. (Figure 2)
4. The folded edge of the patch was sutured with the right ventricular side of the interventricular septum by interrupt mattress suture. The first stitch is sutured at the anterior point of the interventricular septum. The other end is sutured at an appropriate distance to the interventricular septal crest, and passes through the folded autologous pericardium. The appropriate sutured distance is 3-5mm (Figure 3). The stitches are mattress-sutured sequentially until the posterior point of the interventricular septum and transferred to the root of the right AV valve, avoiding the AV node. The suture position should be carefully laid out. The suture method and the method of avoiding the cardiac conduction bundle are consistent with the two-piece method (Figure 3).
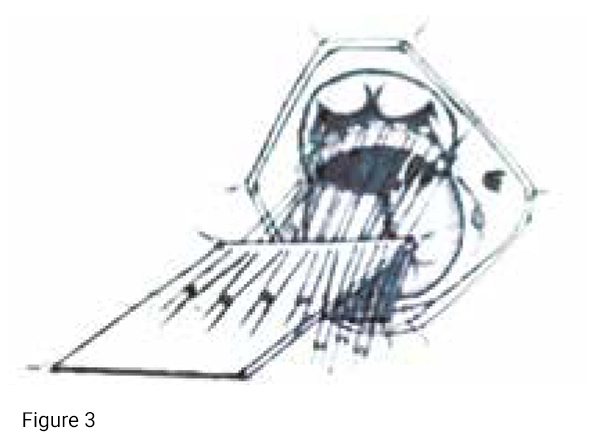
5. Push down the pericardial patch and tie the knot, retract the right pericardial patch or push it into the right ventricle to expose the left pericardial patch and LAVV. Pull the traction thread of the left AV valve to keep the LAVV and the chordae tendineae flat and extended, avoiding the cardiac conduction bundles. Observe the size of the defect between the LAVV and the left interventricular septal crest. Trim the left pericardium patch to an appropriate shape and size. Suture the left pericardial patch and the free edge of the LAVV intermittently or continuously with 7-0 sutures to repair the root defect of the LAVV (Figure 4).
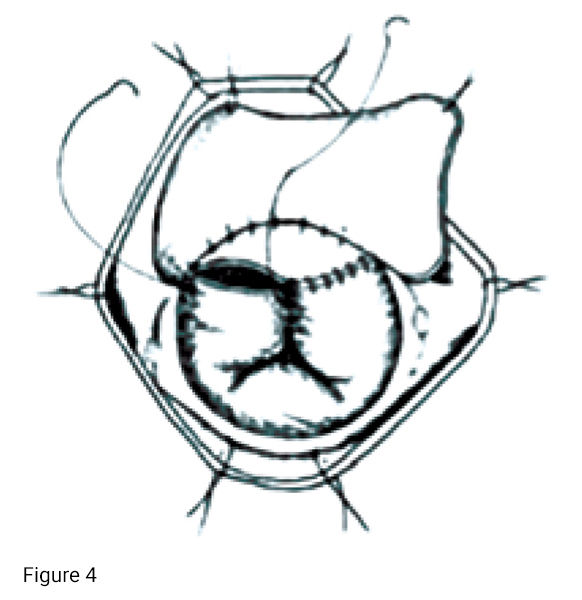
6. Saline solution is injected through the LAVV to test its closure competence. If the LAVV still has central regurgitation and the annular diameter is large (comparing with mitral valve annulus diameters matched to patient’s age and weight), an annuloplasty could be added in the region of the commissure between the LSL and left lateral leaflet and/or in the region of the commissure between the LIL and left lateral leaflet to eliminate LAVV regurgitation.
7. The right patch is also trimmed to an appropriate size and sutured continuously to close the right atrial and ventricular septal defect, separating the coronary sinus into the left atrium. If combined with persistent left superior vena cava, it is necessary to go away from the coronary sinus. The suture method and the method of avoiding the cardiac conduction bundle are the same as the two-piece method.
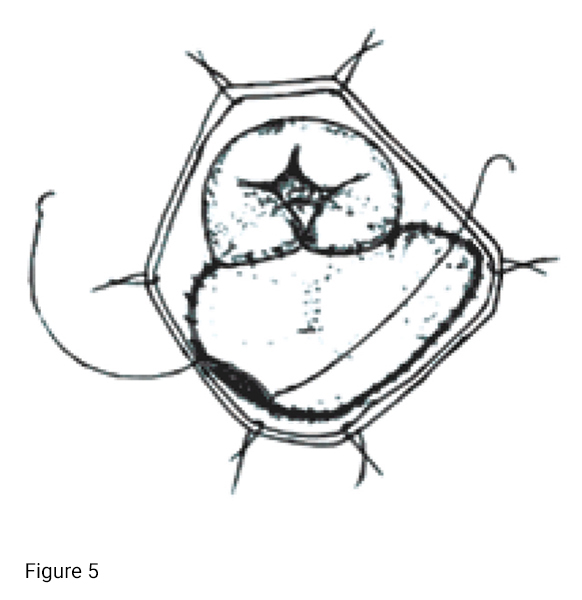
8. Saline solution is injected again to confirm the right AV valve competence after suturing the right superior and inferior leaflets to the appropriate location on the right pericardial patch (Figure 5).
The V-shaped double-layer patch technique is less difficult, eliminating the trouble of accurate measure during the operation. There is no need for special training. The difficulty of the operation is similar to that of the PAVSD technique. The operation time is short, similar to the modified single-patch technique. The V-shaped double-layer patch method is a functional repair surgical technique. The left AV annulus and the right AV annulus are separated to avoid the mutual influence on each other. This technique has maintained the original position, shape and function of the AV valve and subvalvular structure which may reduce the need for reoperation.