Embolic Protection During Carotid Stenting
Using FiberNet device
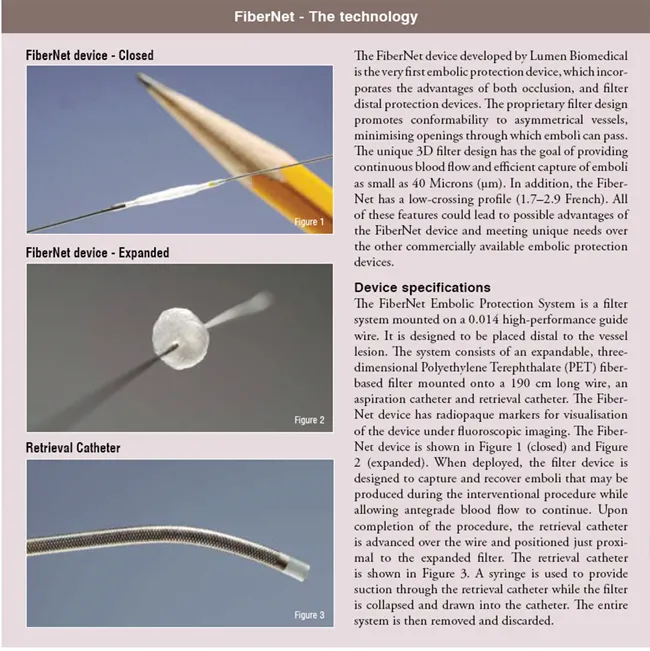
The FiberNet device is the first embolic protection device, which combines features of a filter, and an occlusion device in one system to overcome the complications of carotid angioplasty like distal embolisation of debris.
After having performed angioplasty in coronary and peripheral vascular disease with excellent results, carotid angioplasty was thought to be an opportunity to simplify the treatment of carotid stenosis. Until then, there was no apparent reason to protect patients against distal embolisation. With the development of carotid angioplasty and stenting, prevention of distal embolisation of arteriosclerotic debris and thrombus became a main focus of the intervention. There has been rapid development in new techniques and equipment for carotid artery stenting, including embolic protection devices as adjunctive devices to trap potentially harmful debris that is dislodged during the procedure. Several physicians have reviewed the results for carotid artery stenting with and without embolic protection devices and have shown superior results utilising embolic protection devices during percutaneous interventional procedures. The use of embolic protection devices as an adjunctive device during carotid artery stenting procedure has now become a standard of care. There has been little interest in conducting randomised trials to examine carotid stenting both with and without embolic protection. Most physicians believe that debris that can be seen in the filters is evidence enough. Another explanation is that the complication rate of carotid interventions without embolic protection devices is already low (<5 per cent), which means a large number of procedures would be necessary to prove the benefit of these devices. Currently, there are three types of embolic protection devices for carotid arteries: Distal filter devices, distal occlusion systems and proximal occlusion systems.
- Distal filter devices generally capture only up to a certain size of the particle while allowing continuous blood flow throughout the procedure. Most of these devices have a pore size of 100 microns or larger. Potential disadvantages with filter devices include problematic tracking of the device to the proper location for deployment, the inability to capture small particles, and poor apposition in eccentric or severely diseased vessels.
- Distal occlusion devices interrupt flow to the lesion. The debris from the interventional procedure is aspirated prior to deflating the device. These devices have a low-crossing profile and allow the potential capture of particles less than 100 microns that are dislodged during the procedure. The disadvantage of occlusion devices is the temporary restriction of blood flow in the artery, which may not be well tolerated by patients.
- Proximal occlusion devices interrupt blood flow from the common carotid artery to the lesion along with blocking flow in the external carotid artery. The debris from the interventional procedure is aspirated prior to deflating the device. These devices allow for the potential capture of particles less than 100 microns that are produced from the procedure. Proximal occlusion devices do not require a device to cross the lesion prior to protection. The disadvantage of proximal occlusion devices is, as with the distal occlusion devices, the temporary restriction of blood flow in the artery, which may not be well tolerated by the patient. These devices also require a large introducer sheath and are quite bulky compared to the other available protection devices.
First-in-man trial
The first-in-man trial was reported by M. Henry et al. in June 2007 . Since then, 35 high-grade lesions of the internal carotid artery have been treated in 34 patients by stent implantation under embolic protection with the new FiberNet device. Mean patient age was 71.4 ± 8.8 years (range 50-85 years). 67.6 per cent were male. 29.4 per cent of the lesions were symptomatic.
The procedure was technically successful in 34 / 35 (97 per cent) interventions. There was no stroke or death within 30 days. Two permanent amaurosis and one amaurosis fugax occurred. Visible debris was captured in all cases. No changes were noted in CT/MRI during 30 days post-procedure. The mean surface area of debris caught was 63.8 mm² (range 37.7-107.5mm²).
EPIC European Study
Evaluating the use of the FiberNet Embolic Protection Device in Carotid Artery Stenting
Recently, enrolment in the European multi-centre EPIC study (FiberNetT Emboli Protection Device in Carotid Artery Stenting Study) has been completed. Data presentation is pending. Patients with symptomatic, atherosclerotic stenosis =50 per cent or asymptomatic atherosclerotic stenosis =70 per cent of the internal carotid artery according to NASCET (North American Symptomatic Carotid Endarterectomy Trial) Criteria were included.
NASCET Criteria: Degree of stenosis is calculated from the ratio of the linear luminal diameter of the narrowest segment of the diseased portion of the artery to the diameter of the artery beyond any post-stenotic dilation.
Primary endpoint for this study was defined as all death and stroke within 30 days post-procedure. Secondary end points included:
- Death due to any cause, all stroke, including major, minor, ischemic and non-ischemic and myocardial infarction
- Non-stroke neurological events, such as visual / speech disturbances, amaurosis fugax, confusion, seizure, weakness, and TIA
- Technical success (Successful placement, deployment, and retrieval of the FiberNet device)
- Procedural success (Device success with the absence of any stroke, death, or MI during the hospitalisation)
- Access site complications requiring transfusion or surgical intervention (AV fistula, dissection, hematoma, hemorrhage, pseudoaneurysm, puncture site infection)
Plaque debris analysis
In addition to the feasibility study, plaque debris was collected during randomly selected carotid stenting procedures in which the FiberNet device and other CE marked embolic protection devices were used. Visible debris was captured in all cases. The debris was classified for quantity, analysed histologically and differentiated into atheromatous plaque, cholesterol crystals or macrophage foam cells. Data evaluation is still pending. But, our experience shows that the average mean debris surface area captured with use of the FiberNet device or with proximal occlusion devices was larger than with the use of existing competitive filter devices.
RETRIEVE Feasibility Study
Evaluating the Use of the FiberNet Embolic Protection System in Saphenous Vein Grafts
This is a multi-center prospective study designed to demonstrate the performance and safety of the FiberNet Embolic Protection System when used as an adjunctive device during Saphenous Vein Graft (SVG) intervention. This study will enroll up to 30 participants in up to 10 centres in the United States and Europe. The lesion must be located within the SVG and must be =50 per cent and <100 per cent stenosed. The patient may have one or two SVG grafts to be treated and each SVG may have more than one lesion. The primary objective of the study is to evaluate the safety and performance of the FiberNet Embolic Protection System during SVG interventions. The primary endpoint is Major Adverse Cardiac Events (MACE) rate during 30 days post-procedure. The secondary study objectives are endpoints related to the use of the FiberNet system and additional safety endpoints.
Conclusion
Embolic protection with the FiberNet device is an interesting new concept to prevent peri-procedural stroke in patients with carotid stenosis. Debris may be kept back more efficiently than with other embolic protection devices. To substantiate this theory a larger number of patients treated with competitive filters and proximal protection devices should be analysed.




