The vastly increased complexity of cardiac rhythm therapy over the past several years, demands commensurate improvements in overall device monitoring and telecommunication technology.
In stark opposition to the undeniable demographic changes and medical advancements taking place throughout the world, healthcare payers are increasingly pressurising the treating physicians to reduce their costs. Because of the inherent chronicity of cardiac disease, the costs for managing these patients have risen notably in recent years, while the technological options for the best possible care of the patients have made astounding leaps forward.
Parallel to these demographic and health related changes come significant technological and medical advances in cardiac rhythm therapy. Because of the improvements in device functionality and impressive medical innovation in cardiovascular medicine, the total number of cardiac device patients is rising rapidly. Depending on one's cardiac situation, a patient may receive a standard pacemaker, an implantable cardioverter defibrillator, a device for heart failure-related dyssynchronies or a combination of devices all built into one small implanted device. In addition to the implanted device, the physician must also implant one or more electrical leads, which conduct large and small electrical signals between the microchip device and the heart's chambers virtually each time the heart beats. While most cardiac lead implants are considered permanent hardware in the body, the longevity of the implanted device varies by the patient's cardiac condition and other programmed device functions. Notable improvements in battery technology and more energy-efficient device function, however, have greatly extended the longevity of most devices to several years, at which point the device can typically be safely removed from the patient in the hospital and replaced with a new implanted device during a brief surgical procedure.
For years, a very simple patient-driven telemetry has been available for cardiac device patients. With this decades-old system of data transmission from home, the patient places a magnetised wand over the cardiac device and the stored data are extracted from the device, processed in a small home-based receiver, then transmitted via standard telephone line to the physician's office for appropriate interpretation. However, with the increasing complexity of today's devices and their multiple functions, this rather rudimentary approach to collecting limited device data is losing its appeal. Furthermore, this form of data transmission costs valuable time of physician and clinic staff. It also requires a certain degree of patient involvement, which is typically not preferred when one considers today's complexity of functions and the average age of patient. Clearly, the vastly increased complexity of cardiac rhythm therapy over the past several years, demands commensurate improvements in overall device monitoring and telecommunication technology. Industry has kept pace with these demands for more robust monitoring and data processing. Arguably, the implantable cardiac device manufacturers are at the forefront of highly advanced biomonitoring and implantable sensor technology. Moreover, both Heart Rhythm Society and European Heart Rhythm Association have recently recommended that cardiac rhythm management device manufacturers should develop and utilise wireless and remote monitoring technologies to identify abnormal device behavior as early as possible. To reduce under reporting of device malfunctions, its functional status has to be determined more frequently and more accurately.
An 'intelligent' automated telemonitoring system that communicates bidirectionally back and forth, preferably daily with the implanted device, is required for recognising errant device behaviour, as well as the patient's immediate physiological response to the multitude of programmable therapies these devices now offer. Additionally, device patients frequently experience asymptomatic cardiac-related events, the knowledge of which may be of clinical relevance in appropriate patient care. Some of today's more refined home monitoring systems have the capacity to monitor whether the patient has experienced short periods of tachycardia, atrial fibrillation, inadequate pacing stimuli or a declining heart failure status. Since patients may experience such events completely without pain or in their sleep, a fully-automated system should monitor, review and deliver occasional special reports to the treating physician, so that he or she can respond quickly to problem. If device and patient are functioning as anticipated, the physician does not require a special report. When appropriate, the physician can safely rely on a home monitoring-based follow-up routine for ICD-patients to significantly reduce the follow-up burden in their clinics. An abbreviated mode of patient follow-up was recently confirmed by Brugata, whereby the predictive value of a remote-based device follow-up routine (with one annual follow-up visit) was considered clinically equivalent to standard quarterly patientfollow-ups. The total office follow-up burden for defibrillator devices in this study was reduced by nearly one-half.
In past, physicians would first learn about many critical device malfunctions and physiolgical changes when the patient returned to the clinic for a regularly scheduled follow-up and manual device interrogation, which would only take place two to four times per year, depending on patient status. In one study by Lazarus, the author suggests that widespread cardiac telemonitoring offers information that could allow for the early detection of adverse events two to five months sooner, respectively, than what is currently feasible by using the standard in-office follow-up model, which often tracks device patients quarterly or biannually.

Despite the incredible advances we have witnessed in implantable cardiac device function and reliability over the past several years, it is important to note that even the best functioning cardiac device systems can develop structural defects after days, months or years of use.
Unfortunately, these product failures are very difficult to anticipate and can occur at any given point in the devices' life. Furthermore, the most advanced device system has to be able to adapt immediately to the variances that can arise at any moment within the dynamic human heart. With continuous automatic home monitoring, virtually every imaginable system disturbance can be measured and quickly communicated to the treating physician if the severity of the problem is deemed to be highly relevant clinically.
Today's implanted devices have the potential to monitor many dozens of device parameters and automatically give the physician important early detection information on heart rhythm disturbances, advanced therapy delivery, medication effects and device status via fax, text messaging or a secure website from the doctor's own laptop.
Access to the report data is secure, and physicians have large control over many aspects of early detection reporting and the data management process. Physicians control how, when and where the alerts are sent, so that office staff could also view the reports, if the physician permits such access. By delivering comprehensive data comparable to a standard in-office visit, there is a reduced need for patients to come into the clinic for frequent routine follow-ups, which in turn reduces scheduling strain on the clinic and staff.
The world's first fully automated CRM home monitoring system was developed for commercial application in 2001. Currently, approximately 200,000 device patients are being managed via home monitoring systems worldwide, and this patient population is expected to grow significantly in the coming years with the increased adoption of the technology.
Today's telemonitoring is unique in that standard data transmissions can also be supplemented with special alert messages if the patient or device experiences some clinically urgent functional change. In the event of silent arrhythmias, asymptomatic device and lead changes and delivered shock therapies trigger the telemonitoring to deliver additional special alert messages to the physician via internet, fax or text messaging. Because the triggering and reporting requires no action on the part of the patient or physician, these unique systems are able to recognise and deliver critical messages within a very short time of their actual occurance, as opposed to the old system of standard in-clinic devices follow-up at pre-set quarterly or semi-annual intervals. Although current bidirectional home monitoring systems do not provide for the remote reprogramming of implanted devices, this feature may become reality in the near future.
In recent months, there have been a number of published clinical reports regarding post-implant reliability and malfunction of certain types of defibrillator leads. Current telemonitoring systems make daily painless impedance checks of pacing and shock coils and continually monitor significant changes in these values. Today's device monitoring should be able to quickly and reliably recognise faulty lead performance, providing the highest degree of patient safety and security.
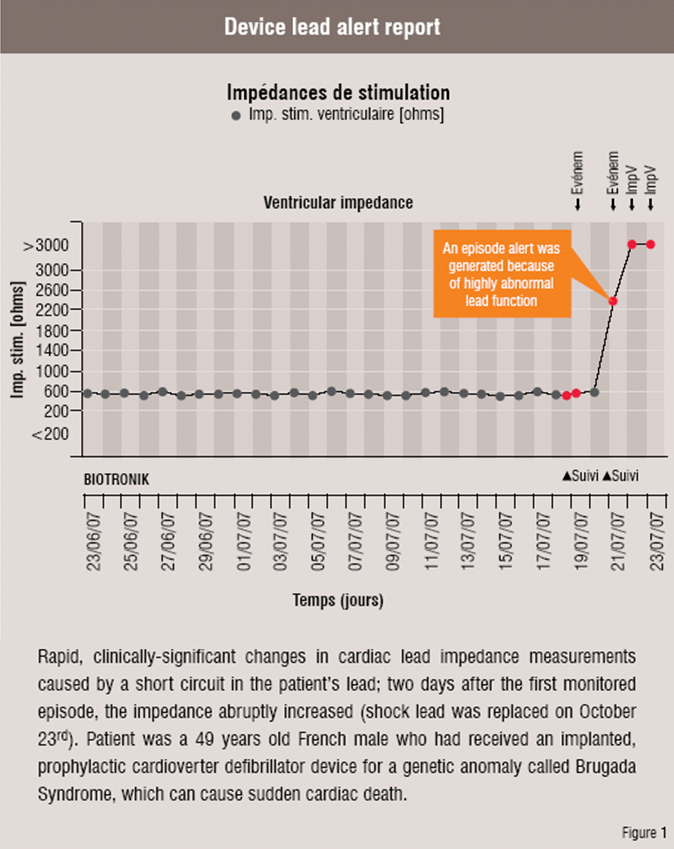
Figure 1 is an example of a device lead alert report generated with one manufacturer's telemonitoring system as soon as a defective shock lead was detected in a middle-aged male.
The real-life situation shown in figure 1 is an example of very early detection of a potentially life-threatening cardiac lead failure. Without an intelligent telemonitoring system, this patient would have retained his defective lead up until the next standard in-clinic follow-up, which could have been months from the date of actual lead failure. Fortunately, the defective lead was quickly identified and immediately replaced within two days once the alert report was automatically sent directly to the treating physician.
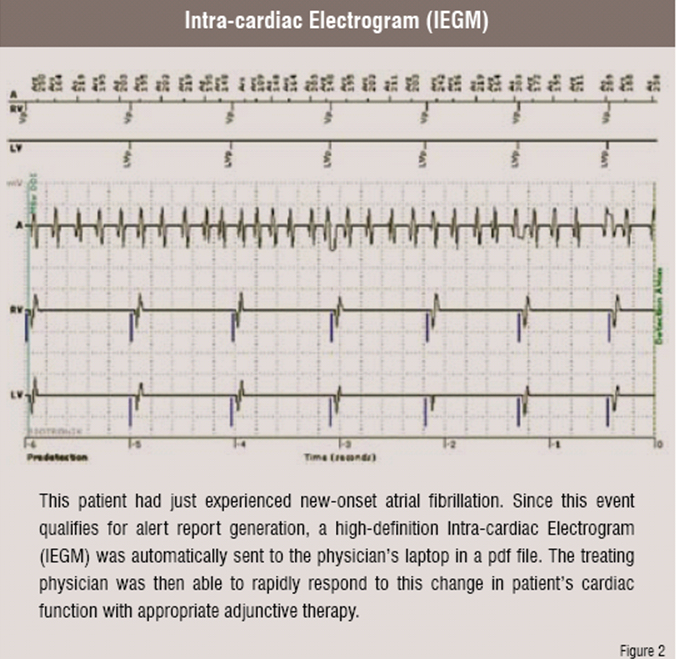
Another example of the value of automated home monitoring is the very early detection of atrial fibrillation, a frequent symptom-free precursor to debilitating embolic strokes and also a common co-morbidity in cardiac device patients. If atrial fibrillation can be recognised early in its course, appropriate medical care-antithrombotic drugs or cardiac ablation-can be initiated to greatly reduce the risk of future strokes. The following Intra-cardiac Electrogram (IEGM) was generated the very day a patient developed atrial fibrillation for a pre-specified duration of time (Figure 2).
For one thing, the automatic monitoring outside the hospital provides clear clinical benefits for the patients, physicians and healthcare providers. Potential medically-relevant problems can be recognised and addressed proactively, avoiding unnecessary hospitalisation costs. The clinical value of such home monitoring systems for cardiac device patients is already well. It appreciated, as is the improved Quality of Life for the patients who are monitored at home daily. This probably is no more apparent than in the management of heart failure patients.
Secondly, the robust incorporation of (cardiac) telemonitoring into a given clinic's work routine will likely require a commitment to redirect certain internal resources and work processes in order to reap the full benefits of a telemetric patient monitoring. Unlike many medical innovations, the true benefits of cardiac telemonitoring will be spread over many of those involved in the patient care cycle.
Finally, a growing number of hospitals and pacemaker clinics have already recognised the inherent value of adopting this new technology to improve work efficiency, deliver more timely urgent medical responses when cardiac events occur, and to use this monitoring tool for establishing a more productive professional interface with a given patient's non-specialist physicians and caregivers.
Essentially, cardiac telemonitoring has very positive near-term growth aspects for a variety of reasons. User-friendly monitoring systems have now been developed by industry and are becoming more prevalent in daily practice across the globe.
Though the clinical advantages of continuous telemonitoring are quite apparent, major ongoing trials are soon expected to more specifically quantify the economic benefits of using this exciting technology. Also, new sensor technology research suggests that numerous physiologic functions might be included in future cardiac monitoring systems, thus widening their applicability.
Because the use of novel technologies also requires a willingness to drop old processes and adopt new ones, clinics and physicians are greeting cardiac telemonitoring with varying degrees of comfort and eagerness. Another structural hurdle is the current lack of reimbursement to the physician or clinic for adopting a system of cardiac telemonitoring.
In countries where healthcare reimbursement for remote monitoring has been instituted, the uptake of this valuable technology has become much more attractive. The US may soon become the model and leader for widespread telemonitoring of cardiac device patients.
In today's advanced telemonitoring systems, the patient's device and physiologic parameters are collected daily throughout the life of the device and are automatically channelled into a series of easy-to-use reports for the physician to review. Standard nightly data transmissions are sent from the implanted cardiac device to a small tabletop receiver, which then sends the encrypted patient and device data via standard telephone line to a central server for immediate processing. Depending on the device manufacturer, individualised patient data are posted in a comprehensive report format with informative trends, charts, parameters, high definition IEGMs and graphs. One device manufacturer also offers a fully mobile, worldwide patient monitoring system via cellular phone network (Biotronik GmbH & Co. KG, Berlin (Germany).